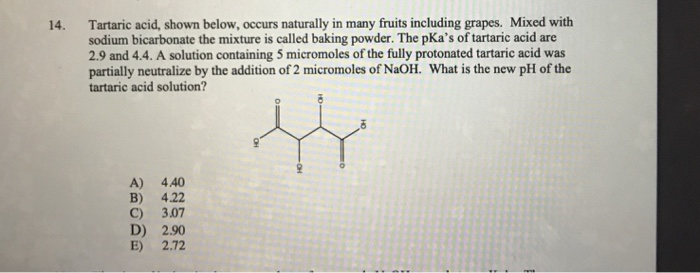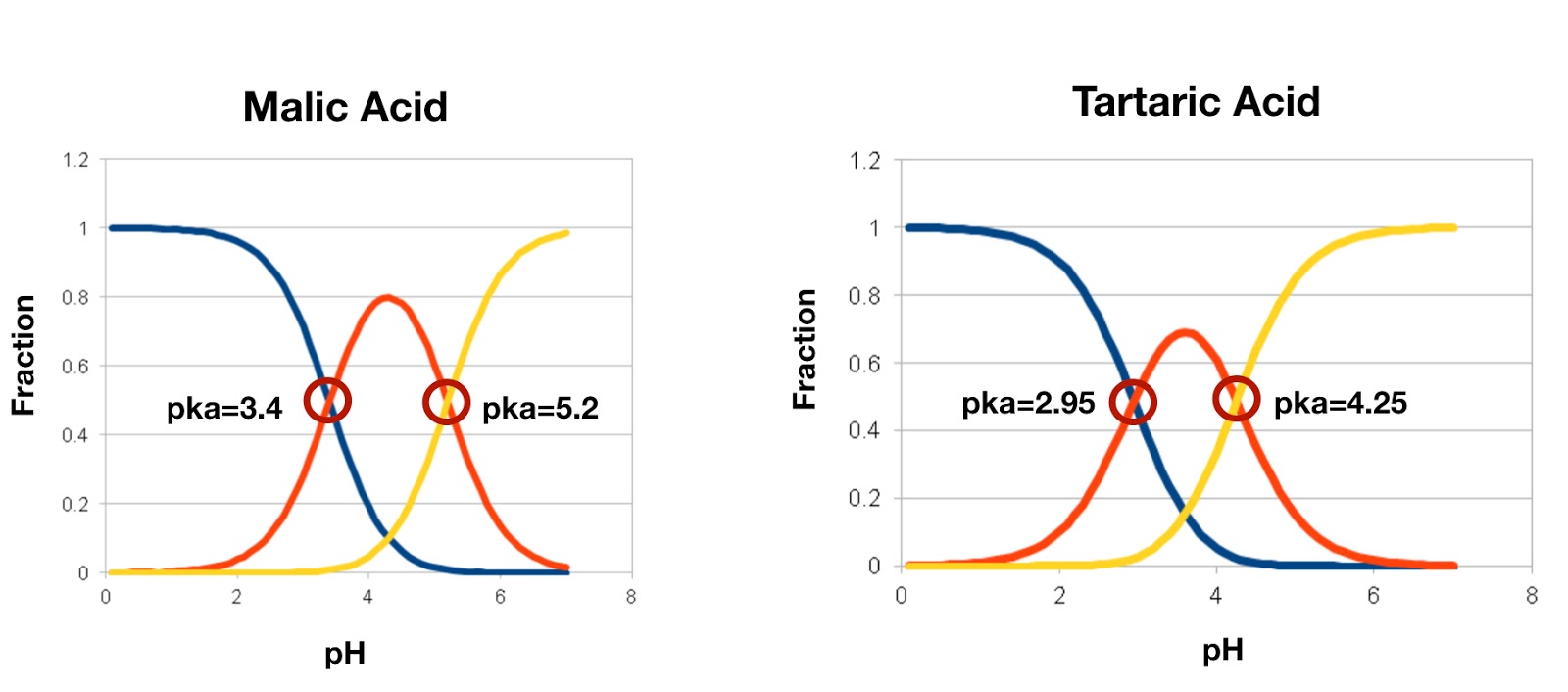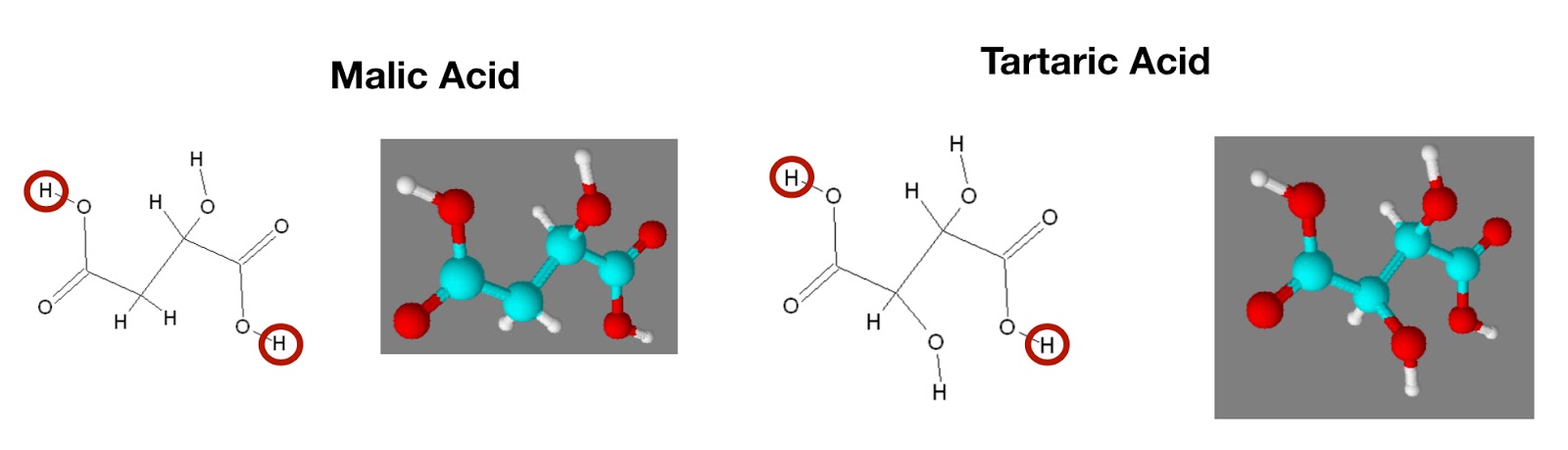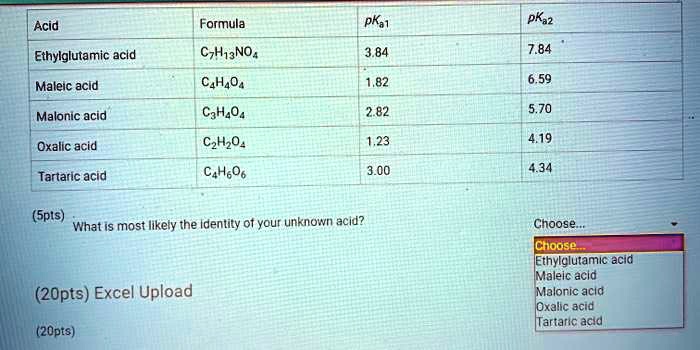
SOLVED: Acid Formula pKa pKaz Ethylglutamic acld CzH13NO4 3.84 7.84 Maleic acid C4H404 6.59 Malonic acid Cat,O4 2.82 5.70 Oxalic acid C2Hz0 1,23 4.19 Tartaric acid C4HsO6 3.00 434 (Spts) What iS
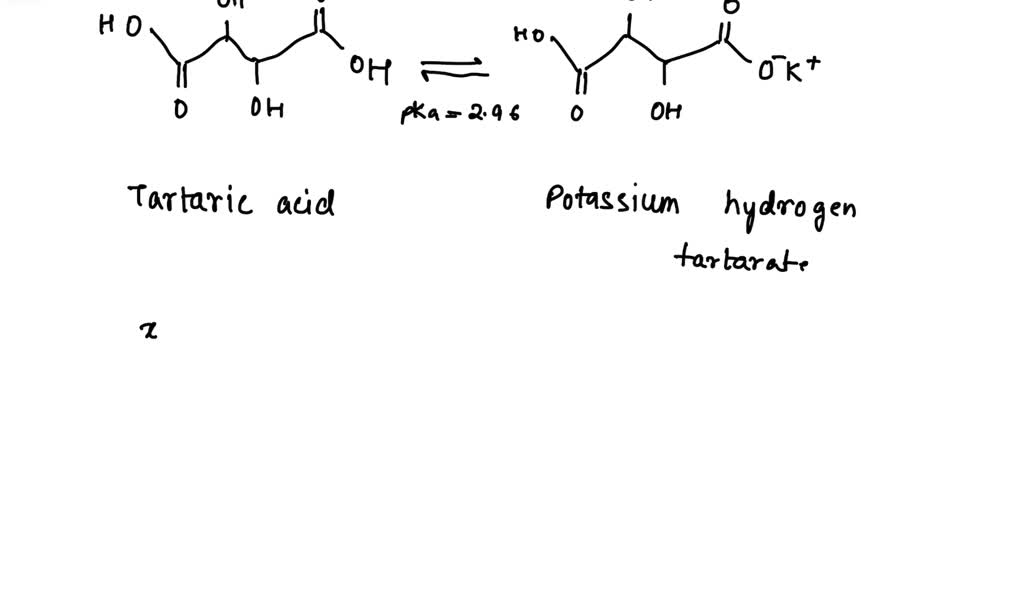
SOLVED: During the fermentation of wine, buffer system consisting of tartaric acid and potassium hydrogen tartrate is produced by a biochemical reaction. Assuming the concentration of potassium hydrogen tartrate is twice that

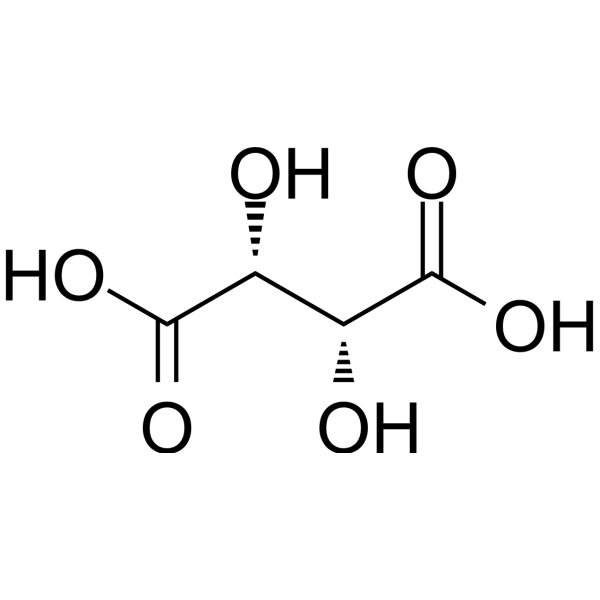
Write all acid-base hydrolysis reactions for the polyprotic acid: d-Tartaric acid. Clearly write the structure of the species, and the charges. Identify all possible acidic and basic species. | Homework.Study.com

SOLVED: Calculate Ka values for the following compounds. a) Tartaric acid ( pKa = 2.98) b) Phenol (pKa = 10.00)