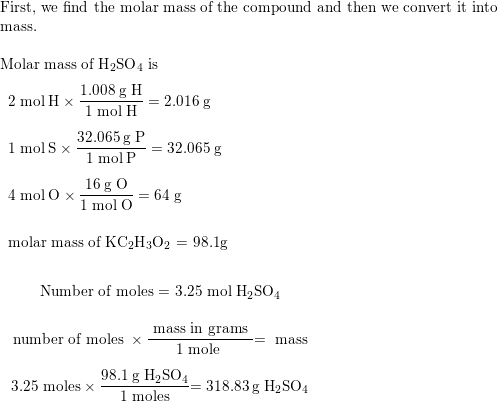SOLVED: 4. What is the molar mass of 3.25 mol of H2SO4 Points) Enter your answer 5. What is the molar mass of 2.5 mol of (C3H5)2 ? Points) Enter your answer

molar mass of sulfuric acid/molecular mass/molecular weight of sulphuric acid/ H2SO4 molar mass. - YouTube

Calculate molecular mass of the following molecules: a. Sulphuric acid (H(2) SO(4)) b. Glucose (C(6) H(12) O(6)) c. Methane (CH(4))
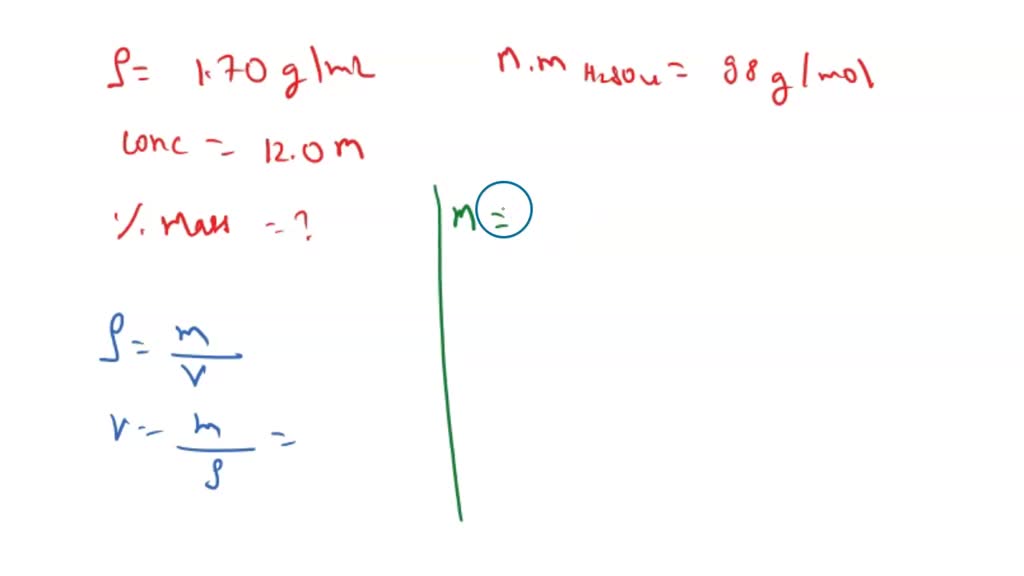
SOLVED: 1. A sample of Sulfuric acid is 12.0 M and has a density of 1.70 g/ml. Find mass % of sulfuric Acid (Molar mass of H2SO4 = 98 g/mol) 2. 250-gram

Question Video: Determining the Concentration of Sulfuric Acid Via Titration with Sodium Carbonate | Nagwa

The molecular mass of H2SO4 is 98 amu. Calculate the number of moles of each elements in 294 g of H2SO4

SOLVED: 9.8 g of H2SO4 is dissolved in enough water to make 500 mL of solution. The molarity of H+ and So ion in the solution respectively are (Assume 100% ionisation of

Campbell / Callis C142B) Chapter #3 : Stoichiometry -Mole - Mass Relationships in Chemical Systems 3.1: The Mole 3.2: Determining the Formula of an Unknown. - ppt download
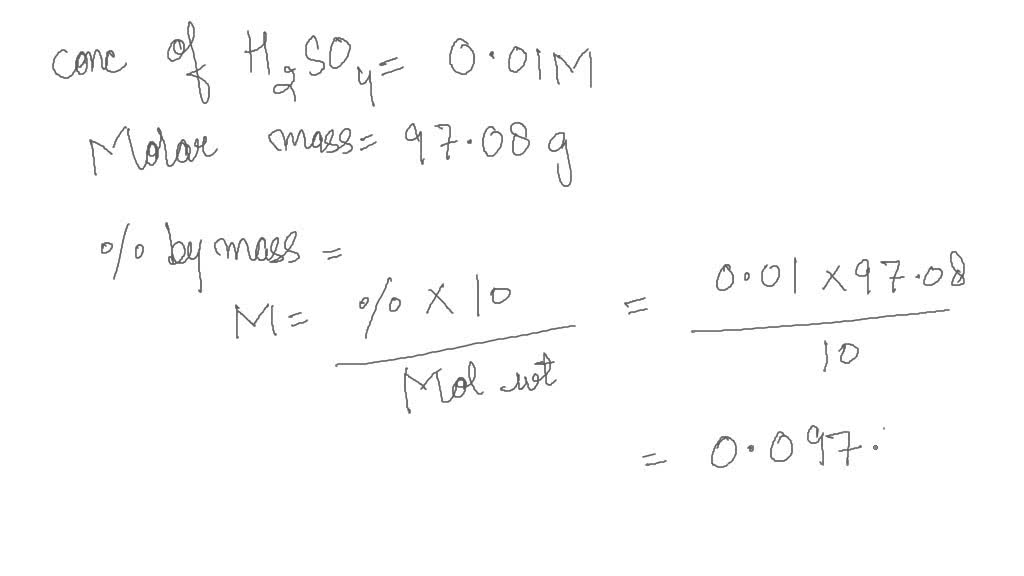
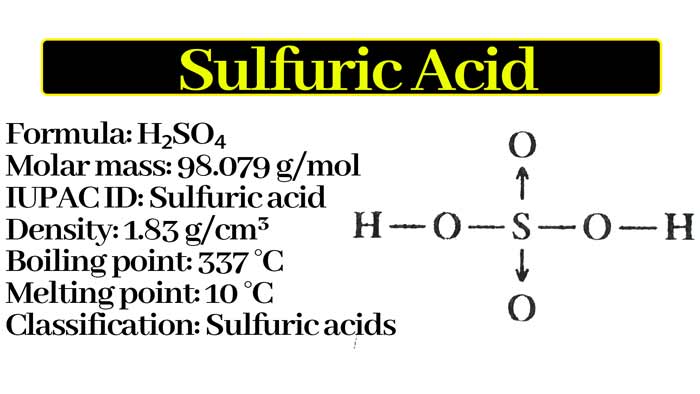

![Sulfuric Acid [H2SO4] Molecular Weight Calculation - Laboratory Notes Sulfuric Acid [H2SO4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/sulfuric-acid-molecular-weight-calculation.jpg)