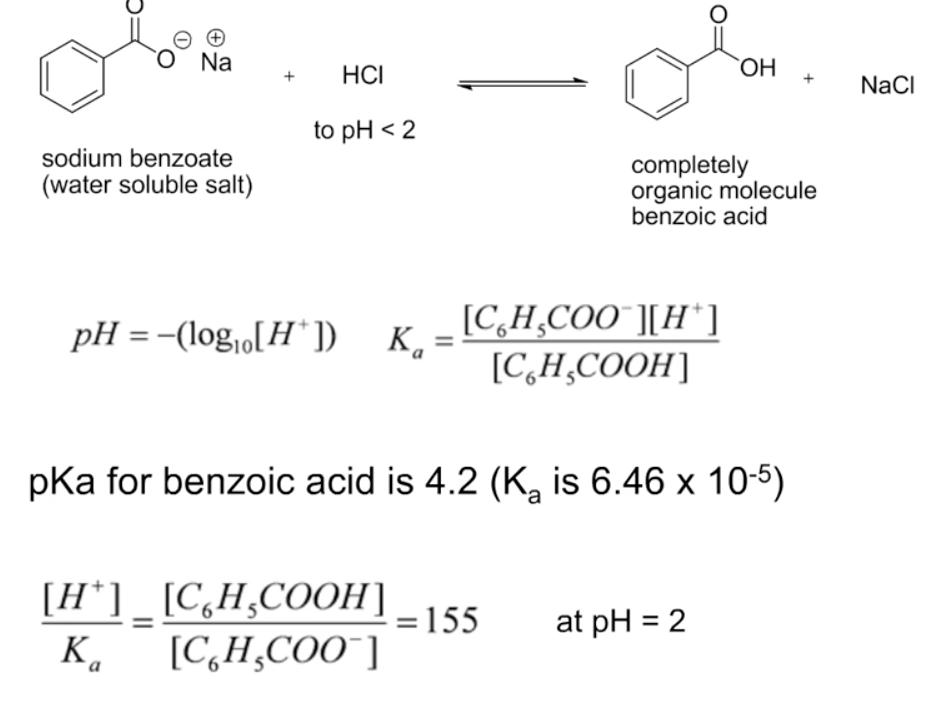
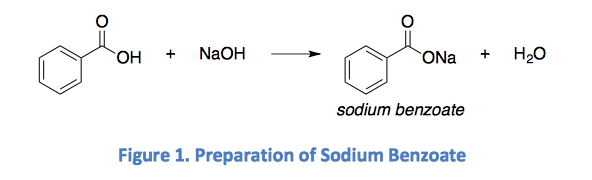
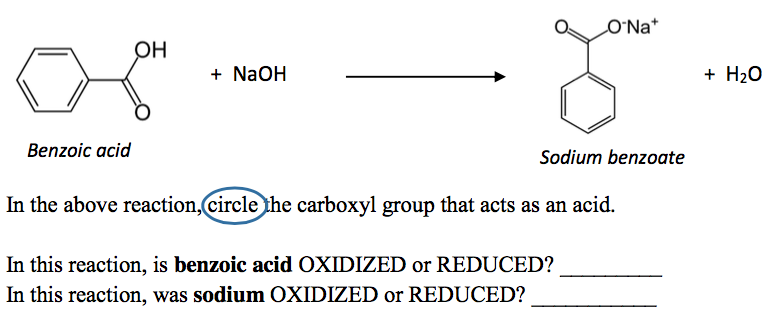SOLVED: You need to prepare 100.0 mL of a pH 4.00 buffer solution using 0.100 M benzoic acid (pKa 4.20) and 0.200 M sodium benzoate. How many milliliters of each solution should

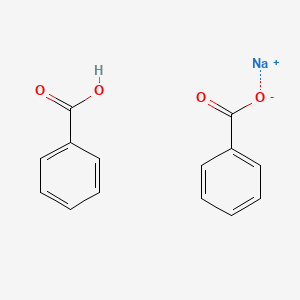
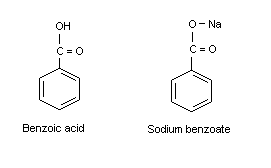
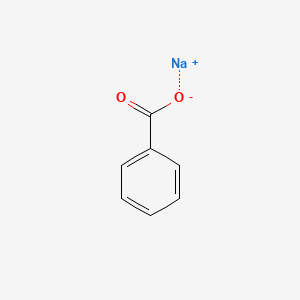
Solved) - Will sodium benzoate be more soluble in water than benzoic acid.... (1 Answer) | Transtutors

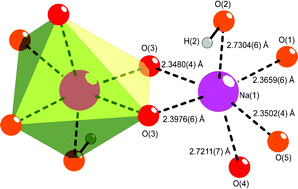
Crystals | Free Full-Text | Co-Crystallization Kinetics of 2:1 Benzoic Acid–Sodium Benzoate Co-Crystal: The Effect of Templating Molecules in a Solution
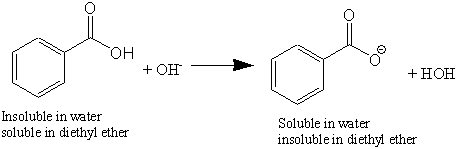
✓ Solved: When benzoic acid ( 5 ) is partitioned between diethyl ether and aqueous sodium hydroxide solution...

Pharmacokinetic data reduce uncertainty in the acceptable daily intake for benzoic acid and its salts - ScienceDirect