Dissociation or Cyclization: Options for a Triad of Radicals Released from Oxime Carbamates | Journal of the American Chemical Society

Oxidation of Primary Amines to Oximes with Molecular Oxygen using 1,1-Diphenyl-2-picrylhydrazyl and WO3/Al2O3 as Catalysts | The Journal of Organic Chemistry

Fragmentation of oxime and silyl oxime ether odd-electron positive ions by the McLafferty rearrangement: new insights on structural factors that promote α,β fragmentation. | Semantic Scholar

Oxime Amides as a Novel Zinc Binding Group in Histone Deacetylase Inhibitors: Synthesis, Biological Activity, and Computational Evaluation | Journal of Medicinal Chemistry

The role of Cinnamomum zeylanicum essential oil, (E)‐cinnamaldehyde and (E)‐cinnamaldehyde oxime in the control of Meloidogyne incognita - Ferreira Barros - 2021 - Journal of Phytopathology - Wiley Online Library

PDF) A newly developed oxime K203 is the most effective reactivator of tabun-inhibited acetylcholinesterase
Chemical Reactivity Measurement and the Predictive Identification of Skin Sensitisers: The Report and Recommendations of ECVAM Workshop 64a - Frank Gerberick, Maja Aleksic, David Basketter, Silvia Casati, Ann-Therese Karlberg, Petra Kern, Ian
![Copper-Catalyzed Tandem [2,3]-Rearrangement and 6π-3-Azatriene Electrocyclization in (E)-O-Propargylic α,β-Unsaturated Oximes | Journal of the American Chemical Society Copper-Catalyzed Tandem [2,3]-Rearrangement and 6π-3-Azatriene Electrocyclization in (E)-O-Propargylic α,β-Unsaturated Oximes | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/ja102436z/asset/images/large/ja-2010-02436z_0004.jpeg)
Copper-Catalyzed Tandem [2,3]-Rearrangement and 6π-3-Azatriene Electrocyclization in (E)-O-Propargylic α,β-Unsaturated Oximes | Journal of the American Chemical Society

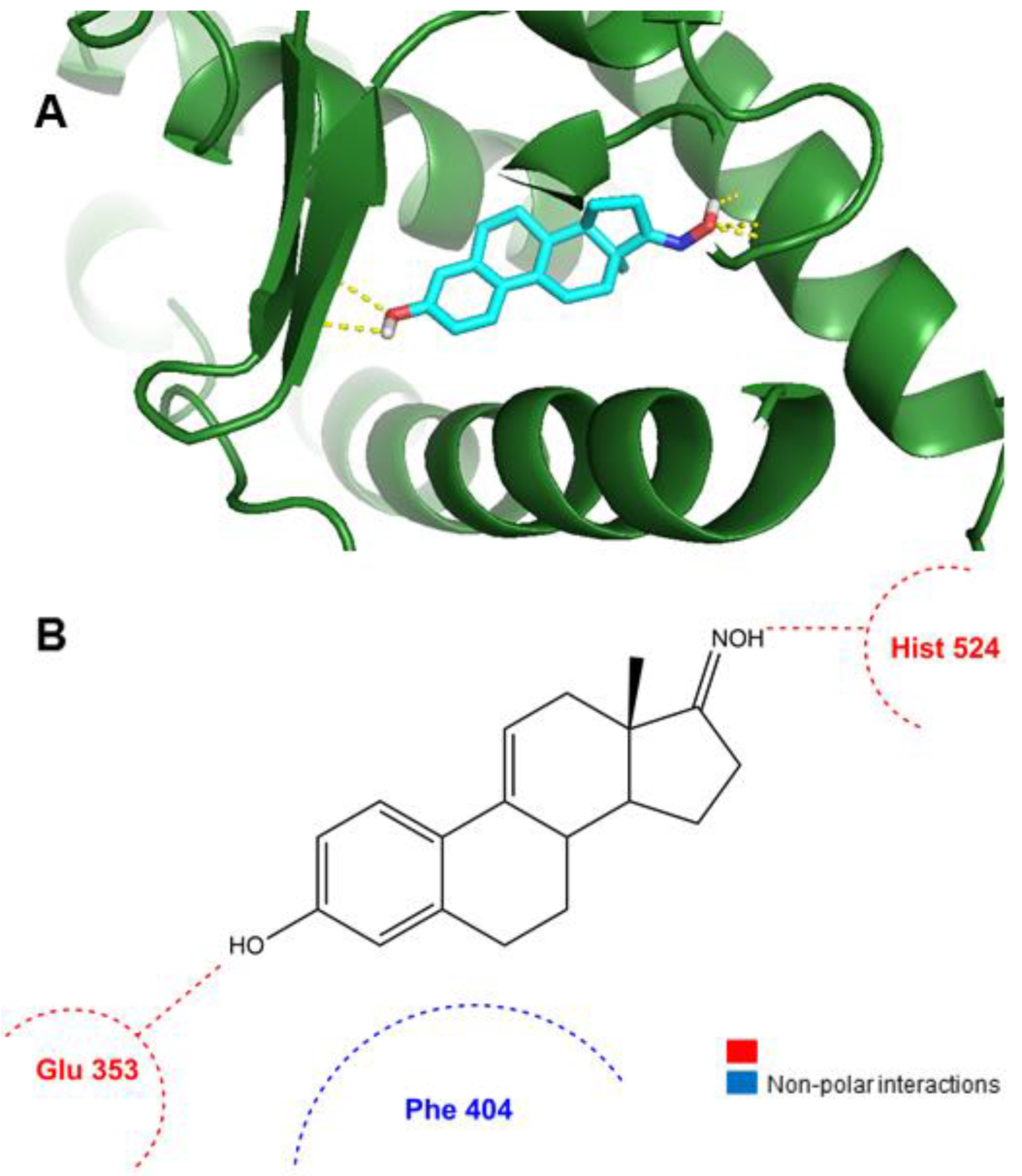
Molecules | Free Full-Text | New Estrone Oxime Derivatives: Synthesis, Cytotoxic Evaluation and Docking Studies

Oxime Amides as a Novel Zinc Binding Group in Histone Deacetylase Inhibitors: Synthesis, Biological Activity, and Computational Evaluation | Journal of Medicinal Chemistry

Typical GC–NICI-MS chromatograms of O-PFB-oxime- TMS derivatives of HNE... | Download Scientific Diagram
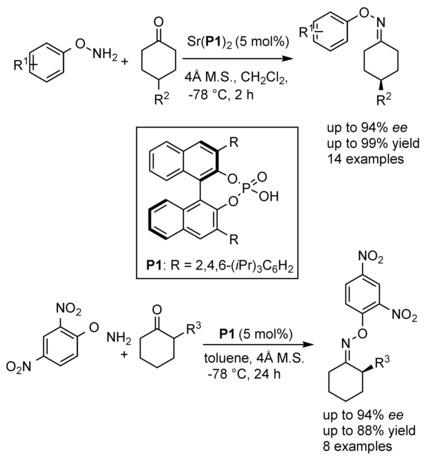
Enantioselective Synthesis of Chiral Oxime Ethers: Desymmetrization and Dynamic Kinetic Resolution of Substituted Cyclohexanones,Angewandte Chemie International Edition - X-MOL

Diastereoselective FeCl3·6H2O/NaBH4 Reduction of Oxime Ether for the Synthesis of β-Lactamase Inhibitor Relebactam | The Journal of Organic Chemistry
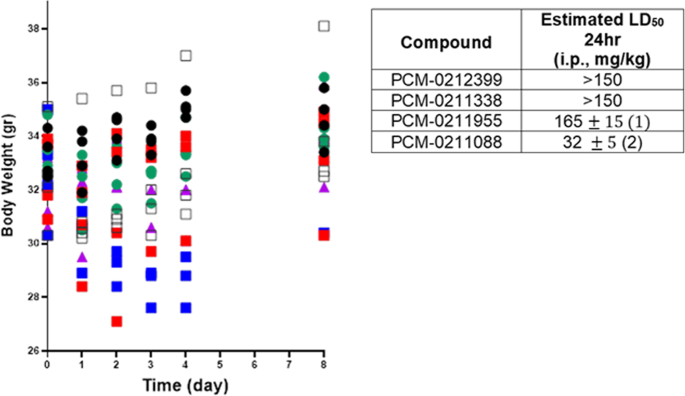
Non-quaternary oximes detoxify nerve agents and reactivate nerve agent-inhibited human butyrylcholinesterase | Communications Biology

Mechanistic Insights into the Ene-Reductase-Catalyzed Promiscuous Reduction of Oximes to Amines | ACS Catalysis
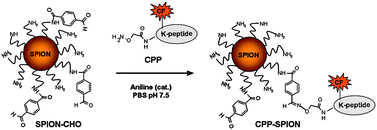
Efficient γ-amino-proline-derived cell penetrating peptide–superparamagnetic iron oxide nanoparticle conjugates via aniline-catalyzed oxime chemistry as bimodal imaging nanoagents - Chemical Communications (RSC Publishing)
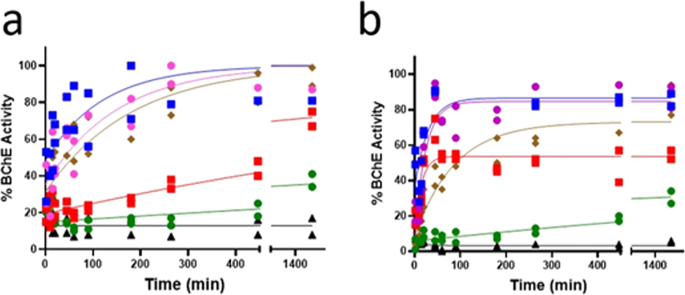
Non-quaternary oximes detoxify nerve agents and reactivate nerve agent-inhibited human butyrylcholinesterase | Communications Biology

Synthesis and Biological Evaluation of d-Amino Acid Oxidase Inhibitors | Journal of Medicinal Chemistry

Live Cell Labeling of Native Intracellular Bacterial Receptors Using Aniline-Catalyzed Oxime Ligation | Journal of the American Chemical Society

Asymmetric Hydrogenation of Oximes Synergistically Assisted by Lewis and Brønsted Acids | Journal of the American Chemical Society