First-in-Class Study Validates Alternate Transport Media and Broader Storage Conditions for SARS-CoV-2 (COVID-19) Testing on RT-PCR Platforms | COPAN Diagnostics Inc.
Service Update: Chlamydia trachomatis and Neisseria gonorrhea – Nucleic Acid Amplification Testing – Updated Process for Sam
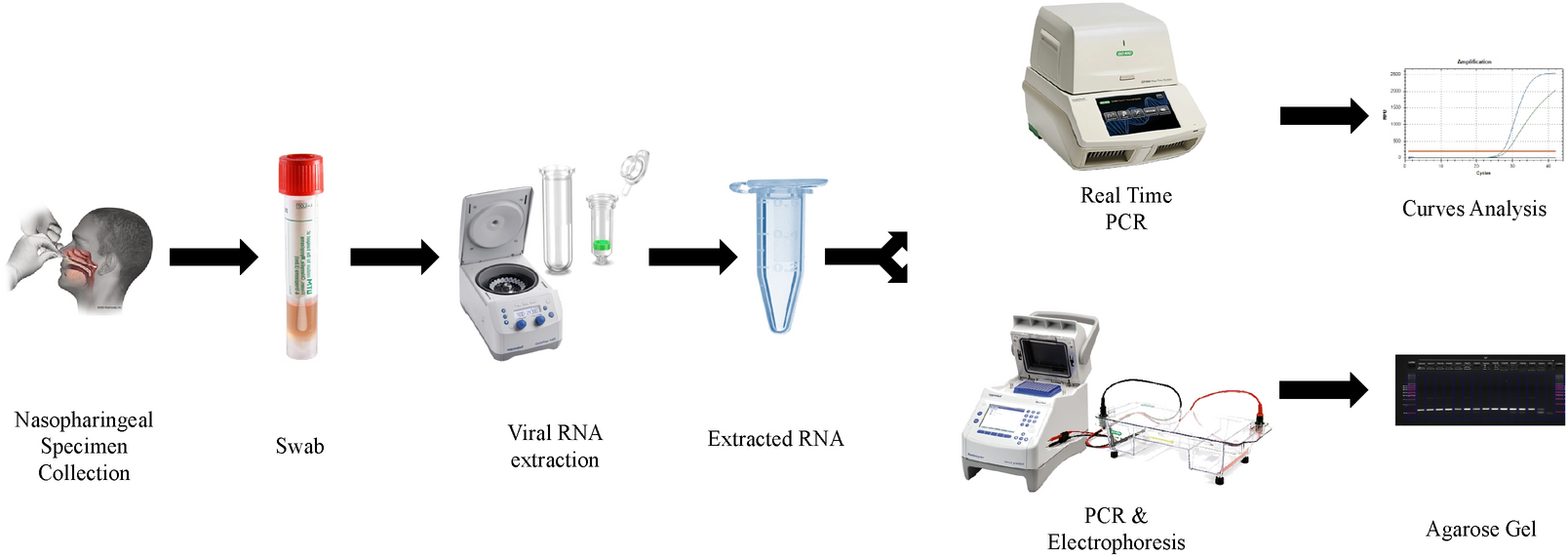
High sensitivity-low cost detection of SARS-CoV-2 by two steps end point RT- PCR with agarose gel electrophoresis visualization | Scientific Reports
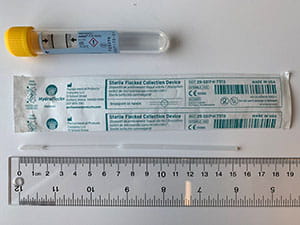
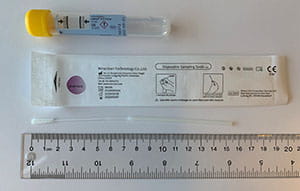
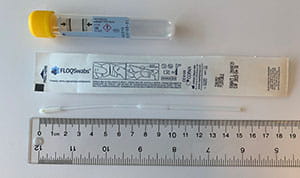
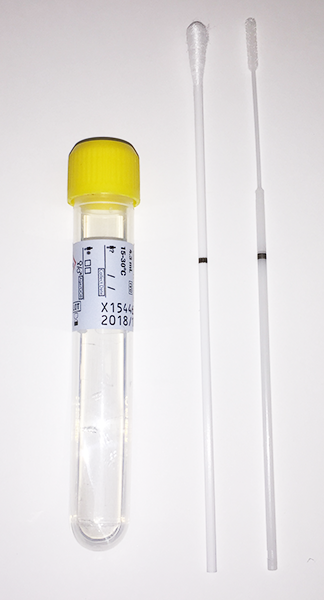
The table below provides guidance on appropriate specimen types based on swab collection kits that may be in your inventory. I