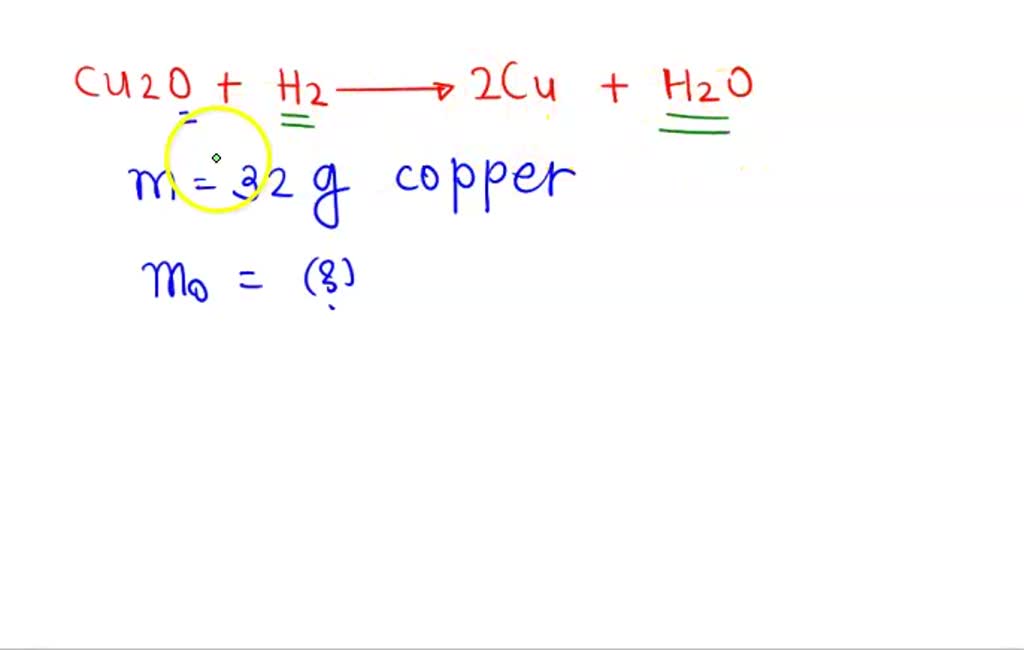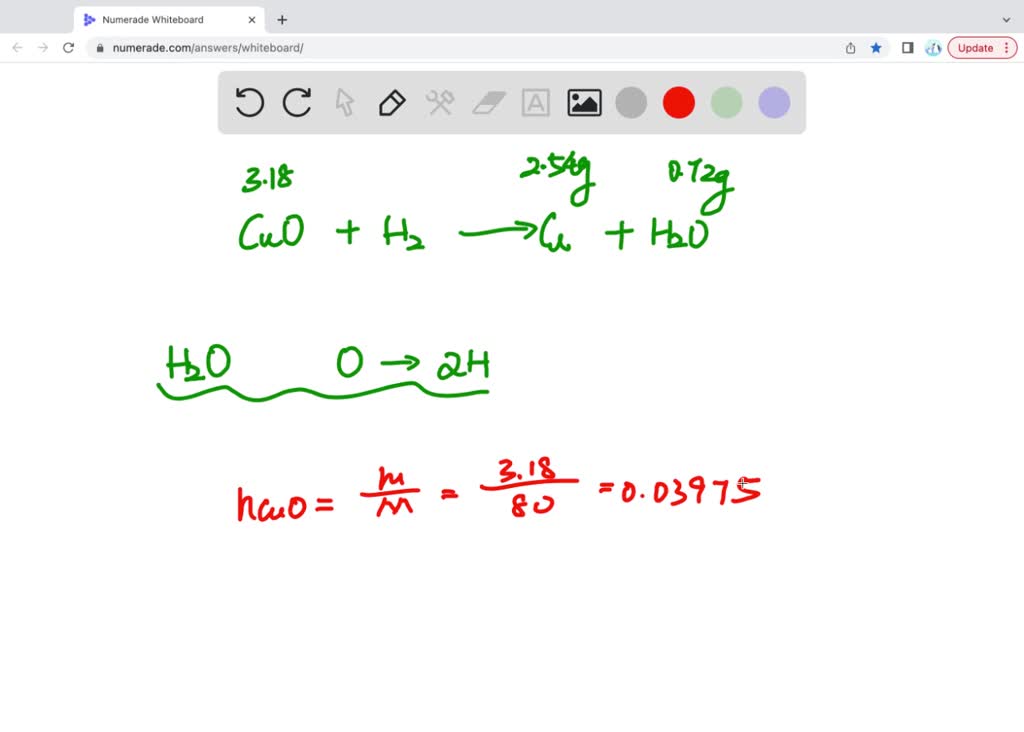
SOLVED: when 3.18 g of copper (||) oxide were carefully heated in a stream of dry hydrogen, 2.54 g of copper and 0.72 g of water were formed determine the number of
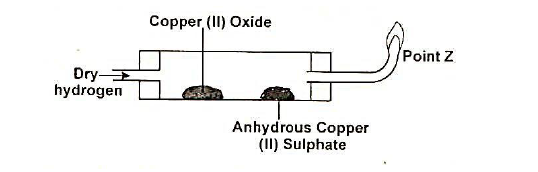
The following diagram was used to study a property of hydrogen gas. Study it and answer the questions that follow.
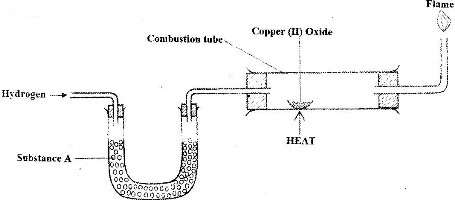
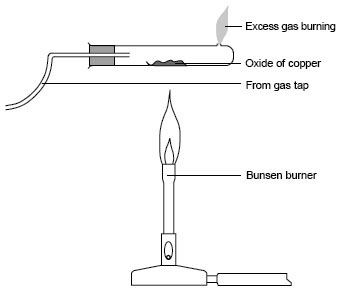
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.

Copper-Modified TiO2 and ZrTiO4: Cu Oxidation State Evolution during Photocatalytic Hydrogen Production | ACS Applied Materials & Interfaces

When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are : {:((i)"elements",(ii),"compounds",(iii),"reactants"),((iv)"products",(v),"metals",(vi),"non ...
When copper (II) oxide is heated in a hard glass tube, and dry hydrogen is passed over, what colour change do you observe? - Quora
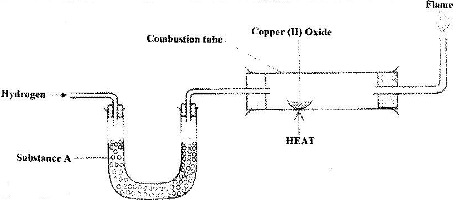
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.
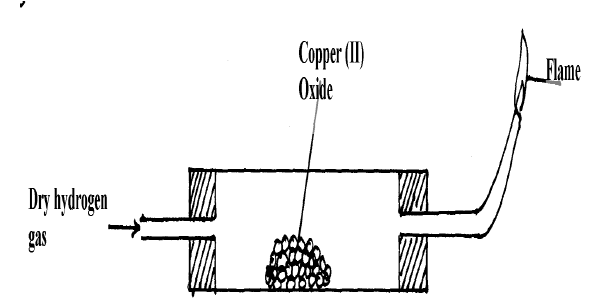
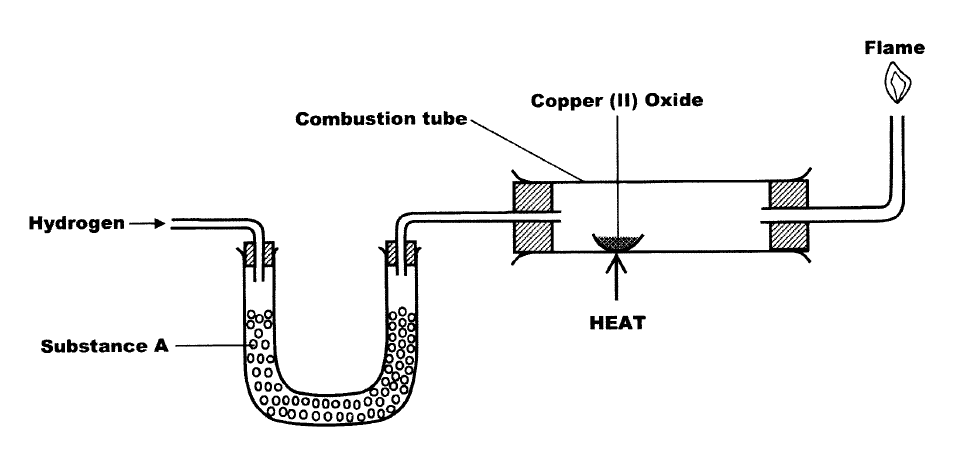
The set-up below is used to investigate the properties of hydrogen. i) On the diagram, indicate what - Tutorke

Morphological changes and kinetic assessment of Cu2O powder reduction by non-thermal hydrogen plasma - ScienceDirect
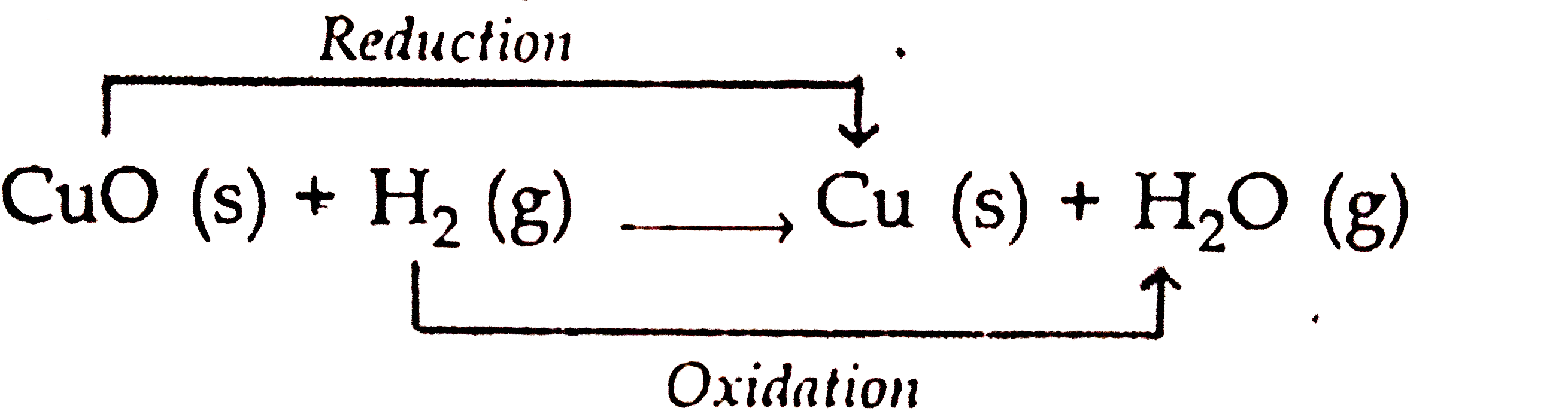
When hydrogen gas is passed over heated copper (II) oxide, copper and steam are formed. Write the balanced chemical equation for this reaction and state (i) the substance oxidized and (ii) the