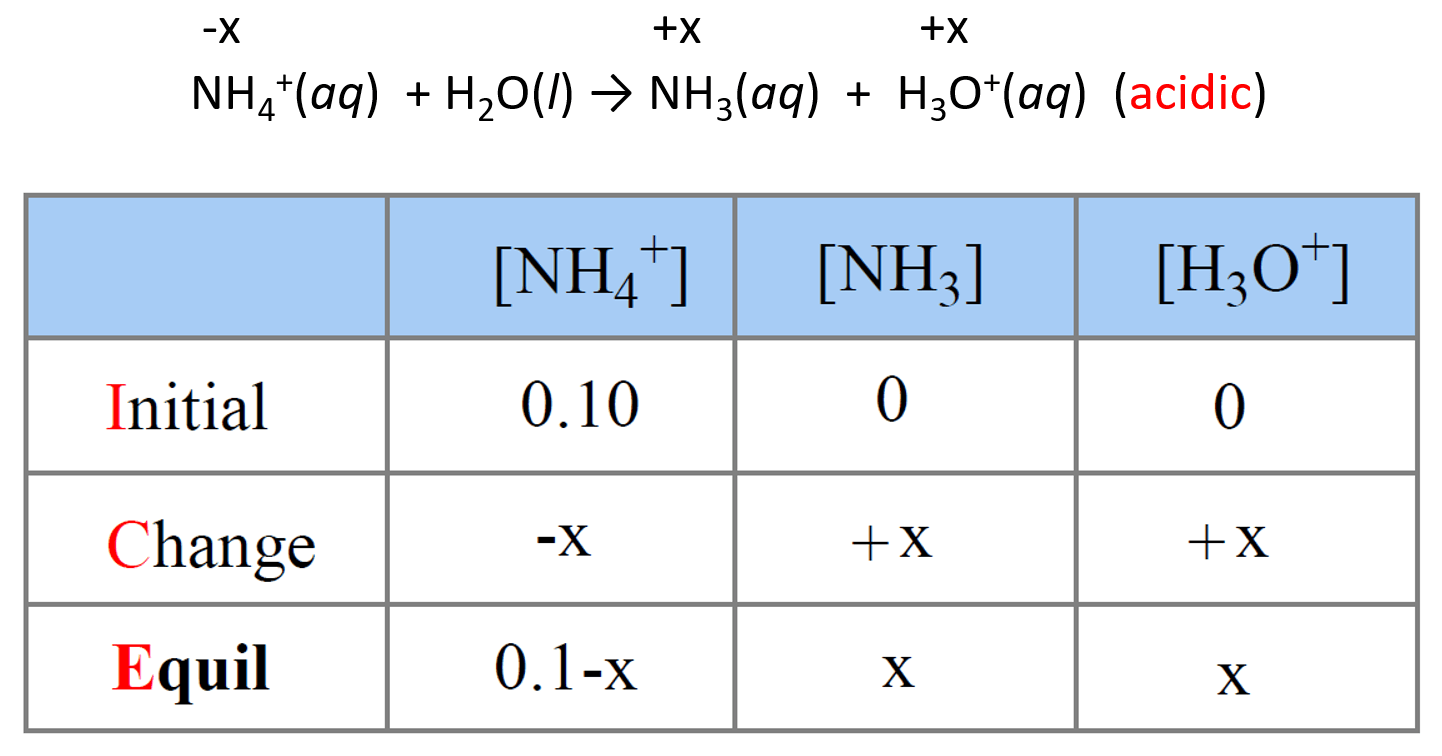
NH3 is a weak base (Kb = 1.8 times 10^-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a solution that is 0.050 M in

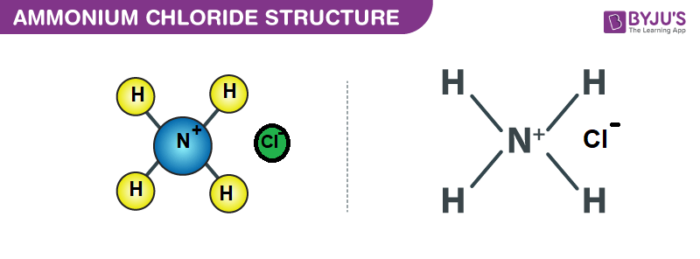
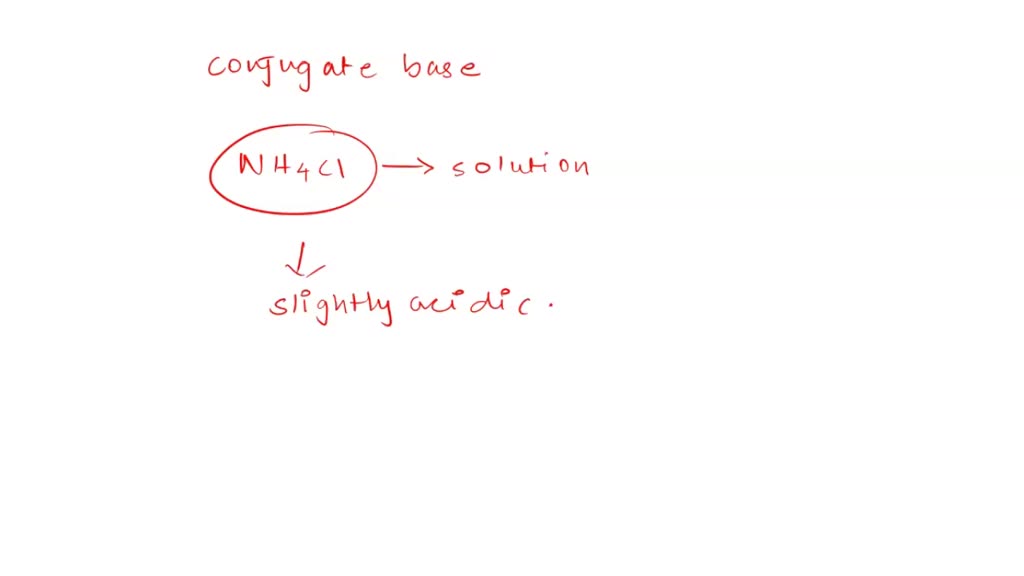
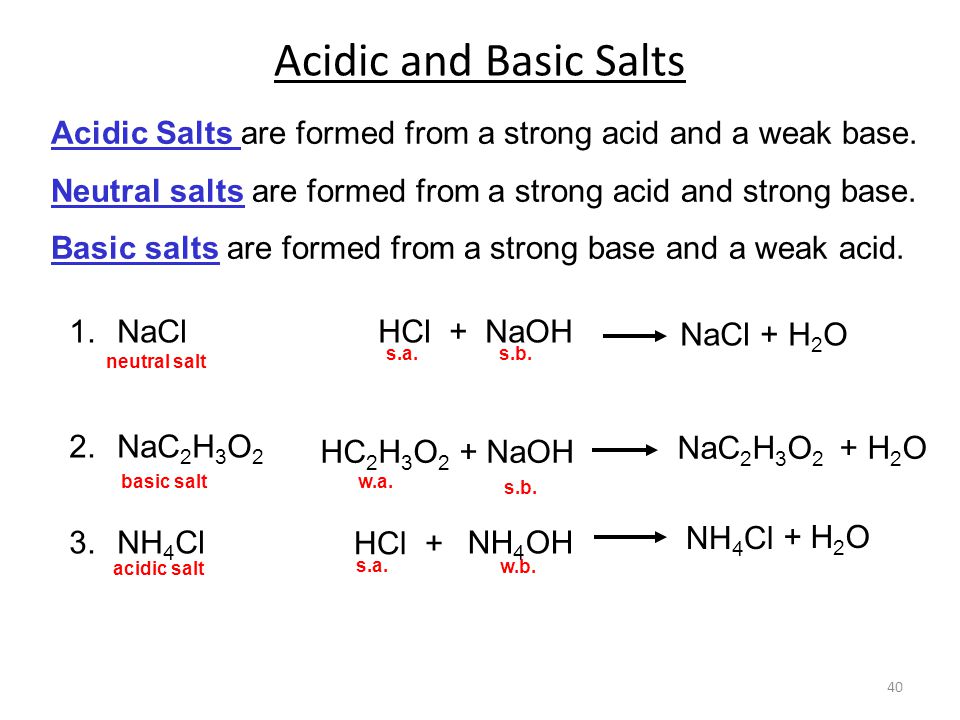
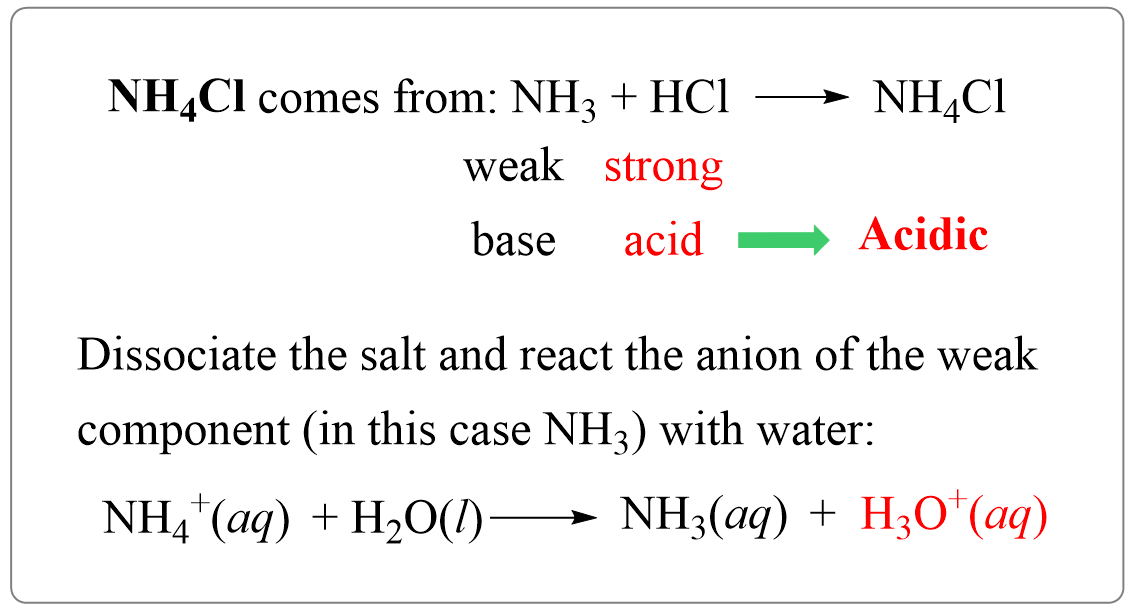
SOLVED: Ammonium chloride (NH4Cl) is an acidic salt. When ammonium chloride is dissolved in water, it dissociates into its component ions. This reaction goes to completion, as indicated by the one-way arrow

Why an aqueous solution of NH4Cl is acidic while that of HCOOK is basic - Chemistry - Ionic Equilibria - 16488273 | Meritnation.com
CHEM 1332 (A.M. Guloy) CHEMICAL EQUILIBRIA--ACID/BASE Acid/base problems may fall into 4 categories: strong acid/base, weak acid