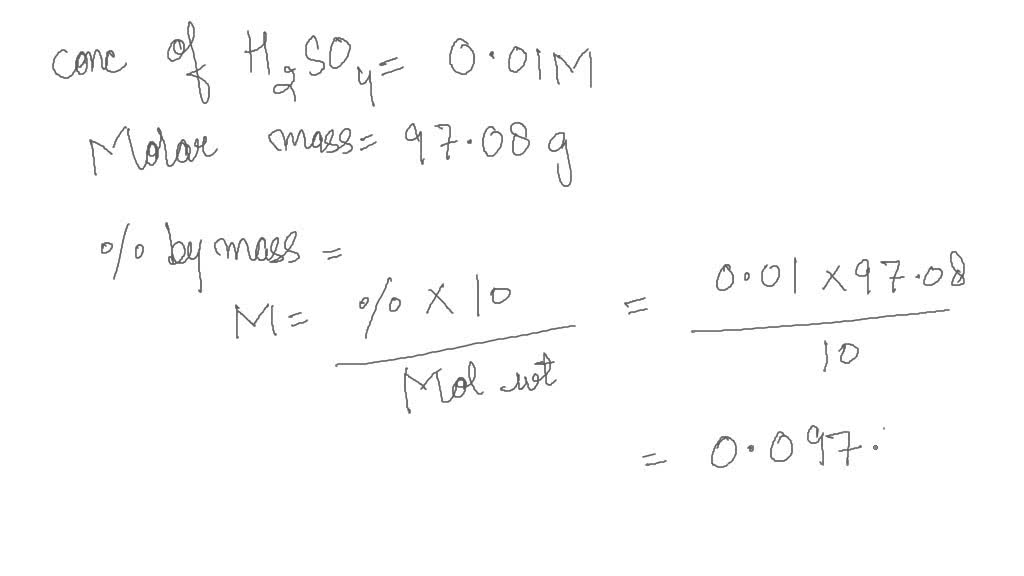
SOLVED: The molarity of a sulfuric acid solution is 0.01 M. Express this concentration in % by mass, ppm, and ppb.(Density of water is 1 g/mL; molar mass of sulfuric acid is

Calculate molecular mass of the following molecules: a. Sulphuric acid (H(2) SO(4)) b. Glucose (C(6) H(12) O(6)) c. Methane (CH(4))

A molecular mass of sulphuric acid is 98 If 49 g of acid is dissolve in water to make - Chemistry - Coordination Compounds - 13542845 | Meritnation.com

SOLVED: A 0.50 molar aqueous solution of sulphuric acid flows into a process unit at a rate of 1.25m3/min. The specific gravity of the solution is 1.03.Calculatea) the mass concentration of H2SO4
The density (in g mL^–1 ) of a 3.60M sulphuric acid solution that is 29% H2SO4 (molar mass = 98g mol^–1 ) by mass will be. - Sarthaks eConnect | Largest Online Education Community

SOLVED: Q2 m3 A 0.5-molar aqueous solution of sulphuric acid flows into process unit at rate of 1.25 min: The specific gravity of the solution is 1.03 and the molar mass of

Calculate the mass of 1 5 grams molecules of sulphuric acid - Chemistry - Some Basic Concepts of Chemistry - 15496931 | Meritnation.com

Calculate the mass percent `(w//w)` of sulphuric acid in a solution prepared by dissovles 4 g of - YouTube
How to calculate molarity and molality of a 14% solution (by weight) of H₂SO₄ (density of H₂SO₄=1.1 g/cm³) - Quora

Calculate molecular mass of the following molecules: a. Sulphuric acid `(H_(2) SO_(4))` b. Gluco... - YouTube














