
SOLVED: First Reduction Step (This first step is investigated second in the video) #4). What is the balanced reduction half reaction of MnOcto MnO4? in basic conditions? MnO4 2- MnOt + e-
![Solid-Phase Quasi-Intramolecular Redox Reaction of [Ag(NH3)2]MnO4: An Easy Way to Prepare Pure AgMnO2 | Inorganic Chemistry Solid-Phase Quasi-Intramolecular Redox Reaction of [Ag(NH3)2]MnO4: An Easy Way to Prepare Pure AgMnO2 | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/acs.inorgchem.0c03498/asset/images/medium/ic0c03498_0011.gif)
Solid-Phase Quasi-Intramolecular Redox Reaction of [Ag(NH3)2]MnO4: An Easy Way to Prepare Pure AgMnO2 | Inorganic Chemistry

Balance redox reaction by ion electron method or half reaction method. Mn2++BiO3-+H+=MnO4-+Bi3+ - YouTube
![SOLVED: Consider the following unbalanced redox reaction Cu'(aq) (MnOa) (aq) 7 > Cu?* (aq) Mn2* (aq) A) identify the oxidation state of the following elements Cu in Cu" is [Select ] Mnin SOLVED: Consider the following unbalanced redox reaction Cu'(aq) (MnOa) (aq) 7 > Cu?* (aq) Mn2* (aq) A) identify the oxidation state of the following elements Cu in Cu" is [Select ] Mnin](https://cdn.numerade.com/ask_images/73ca61cc07fe48df942e93bc7bfee015.jpg)
SOLVED: Consider the following unbalanced redox reaction Cu'(aq) (MnOa) (aq) 7 > Cu?* (aq) Mn2* (aq) A) identify the oxidation state of the following elements Cu in Cu" is [Select ] Mnin
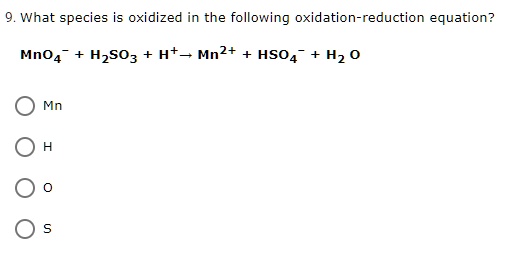
SOLVED: What species is oxidized in the following oxidation-reduction equation? Mno4 H2SO3 H+ Mn2+ HSO4 H2 0 Mn

Balance the redox reaction by ion electron method or half reaction method. MnO4-+SO3-2=MnO2+SO4-2. - YouTube
![SOLVED: Balance the following oxidation-reduction reaction which occurs in acidic solution. Al(s) + MnO4-(aq) <–> MnO2(s) + Al(OH)4-(aq) [Please attach a file showing your work to balance this redox reaction.] SOLVED: Balance the following oxidation-reduction reaction which occurs in acidic solution. Al(s) + MnO4-(aq) <–> MnO2(s) + Al(OH)4-(aq) [Please attach a file showing your work to balance this redox reaction.]](https://cdn.numerade.com/ask_previews/bbae21cb-d160-4e91-b94c-585b639fdfa7_large.jpg)




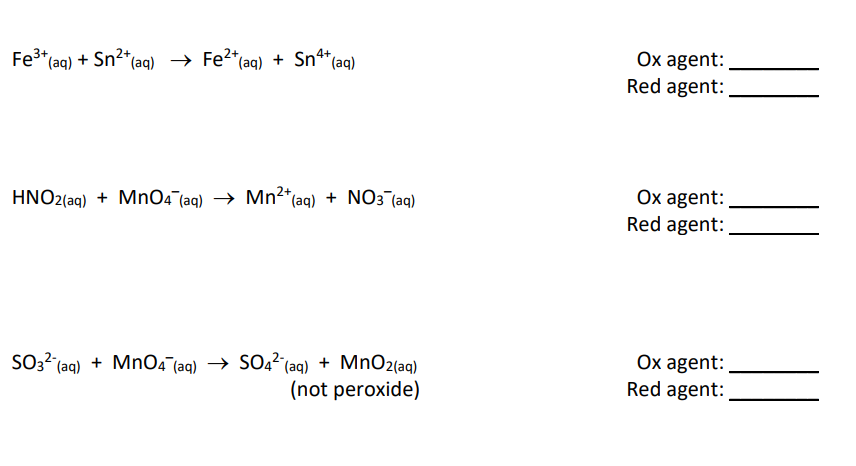




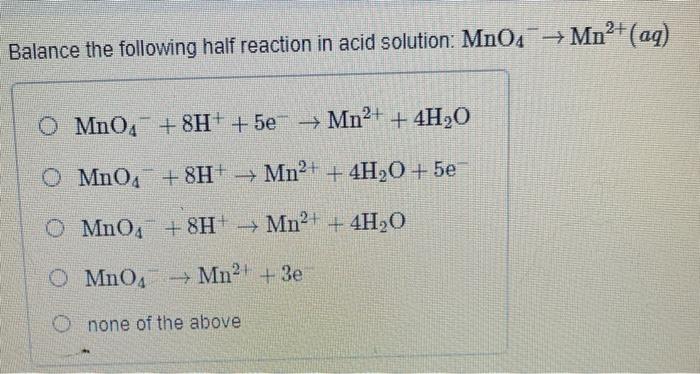


![Interconversion scheme of permanganate complexes [8]. | Download Scientific Diagram Interconversion scheme of permanganate complexes [8]. | Download Scientific Diagram](https://www.researchgate.net/publication/355849905/figure/fig1/AS:1085746865803265@1635873633207/Interconversion-scheme-of-permanganate-complexes-8.jpg)

