Pharmaceutics | Free Full-Text | BX795-Organic Acid Coevaporates: Evaluation of Solid-State Characteristics, In Vitro Cytocompatibility and In Vitro Activity against HSV-1 and HSV-2

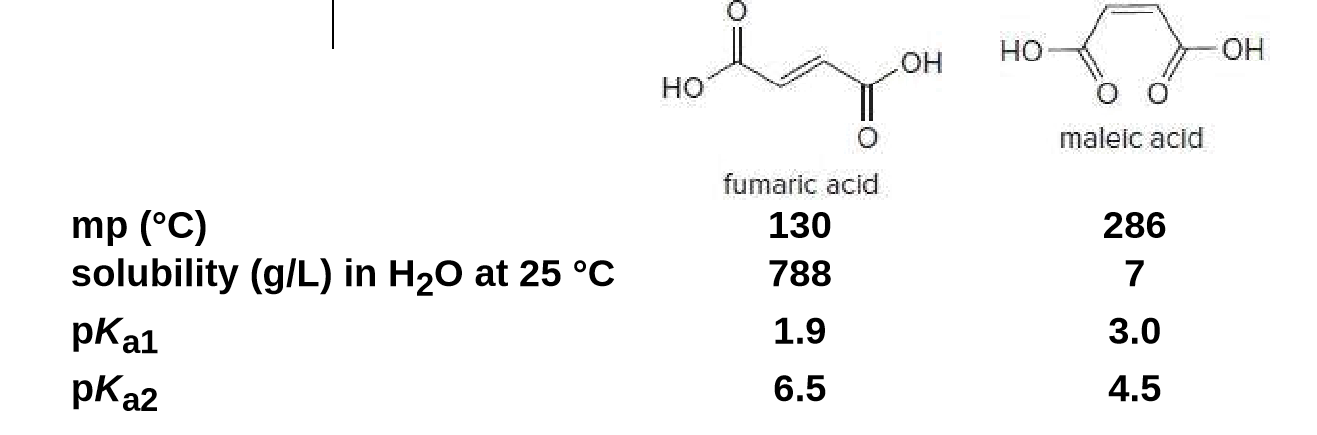
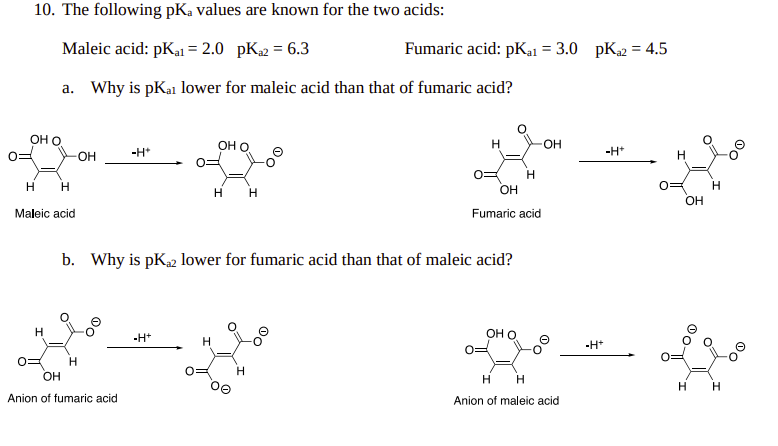
There is restricted rotation around carbon-carbon double bonds. Maleic acid and fumaric acid are two isomers with vastly different physical properties and pKa values for the loss of both protons. Explain why

Scheme 1 Perturbation of pK a values in model dicarboxylic acid systems... | Download Scientific Diagram

SOLVED: Understand: Maleic acid contains two carboxylic acid groups, one with a pKa 0f2.0 and a second with a pKa of 4.0. The fully protonated form of maleic acid is shown on
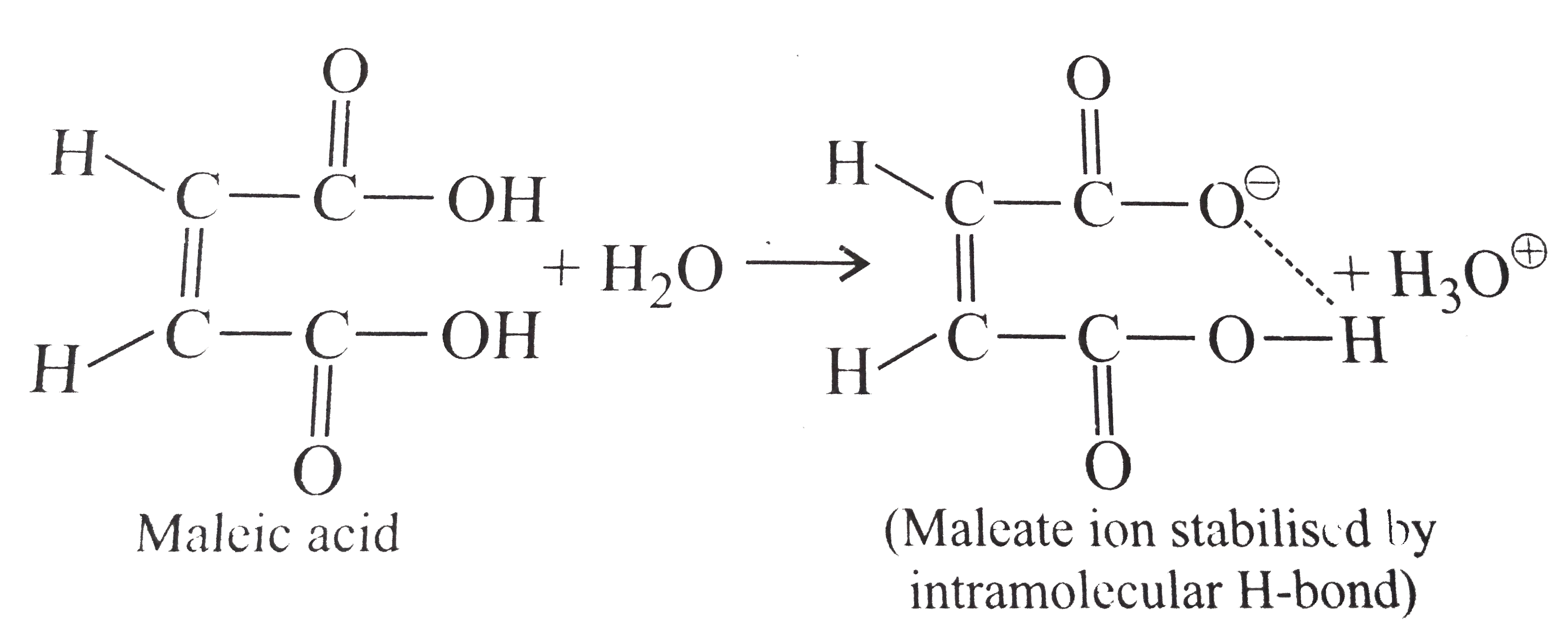
Explain the following : (a) Highly branched acids such as are less acidic than unbranched acids. (b) K(a1) for maleic acid is greater than that for fumaric acid or pK(a1) for maleic