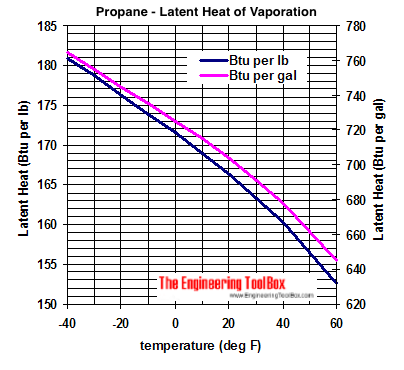
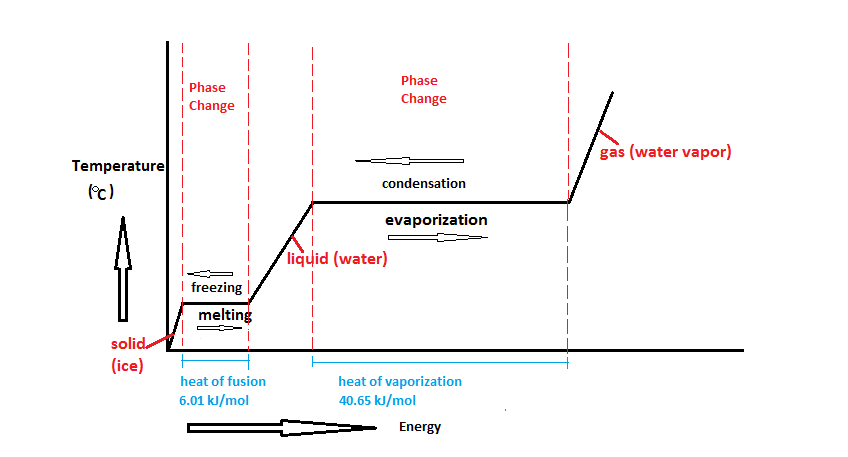
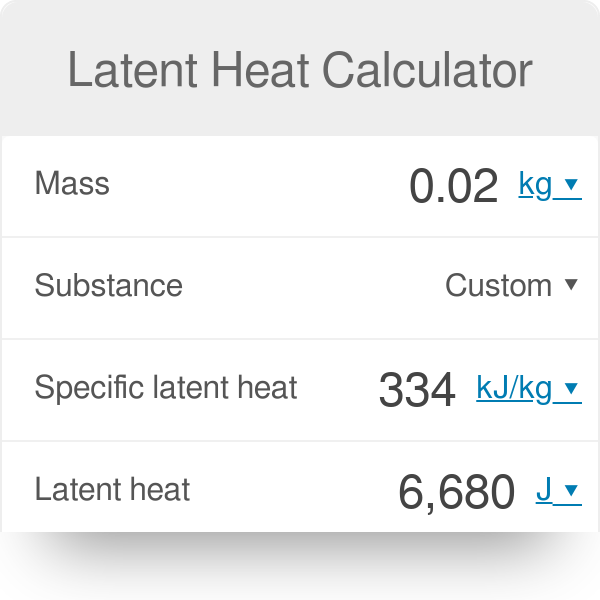
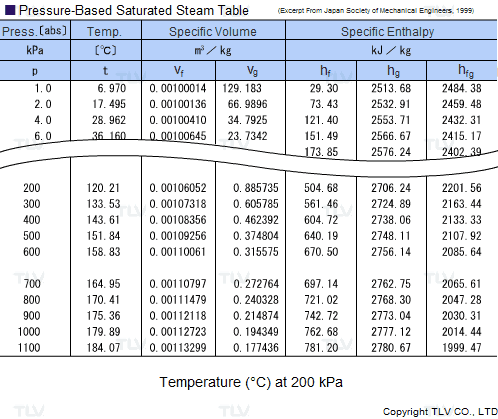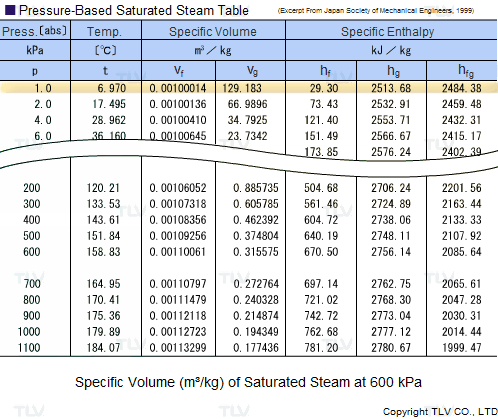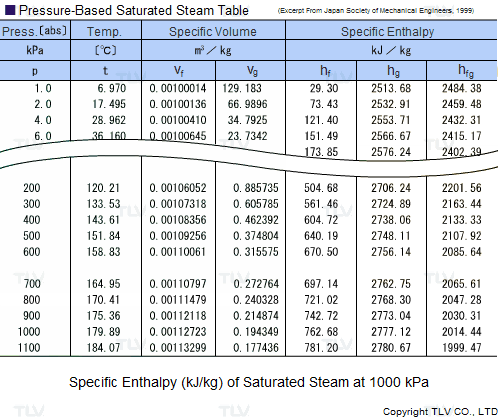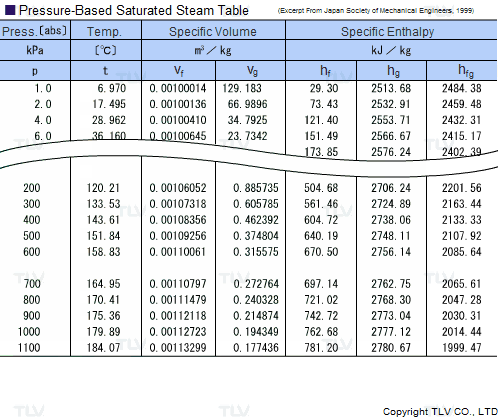
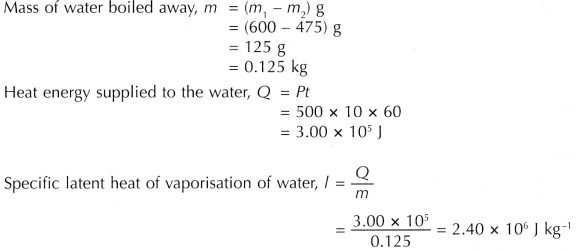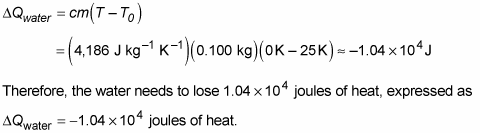Calculate the heat required to convert 3 kg of ice at -12^(@)C kept in a calorimeter to steam at 100^(@)C at atmospheric pressure. Given, specific heat capacity of ice = 2100 J
Calculate the molal elevation constant of water if molar enthalpy of vaporisation of water at 373 K is 40.585 kJ/mol.