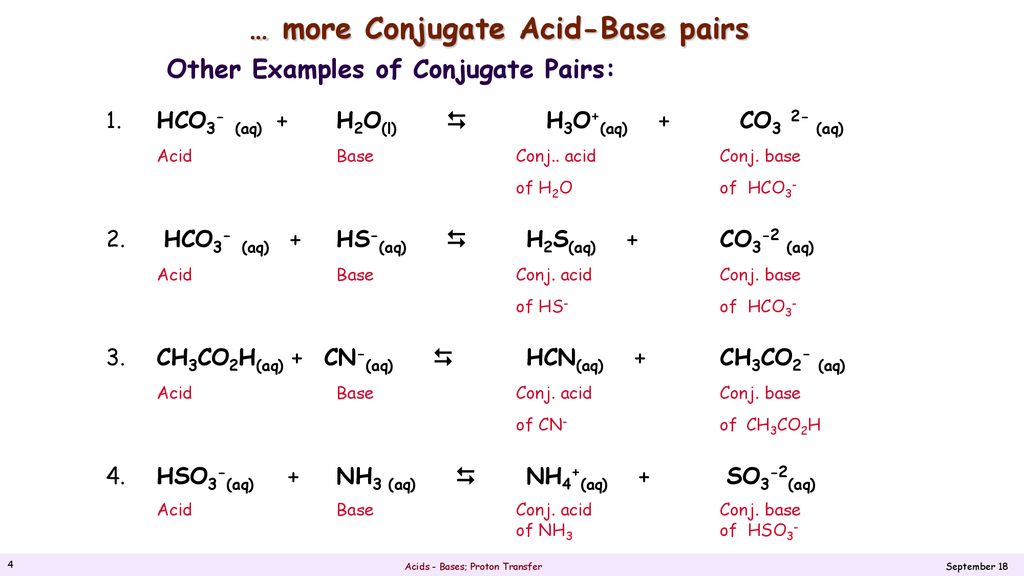
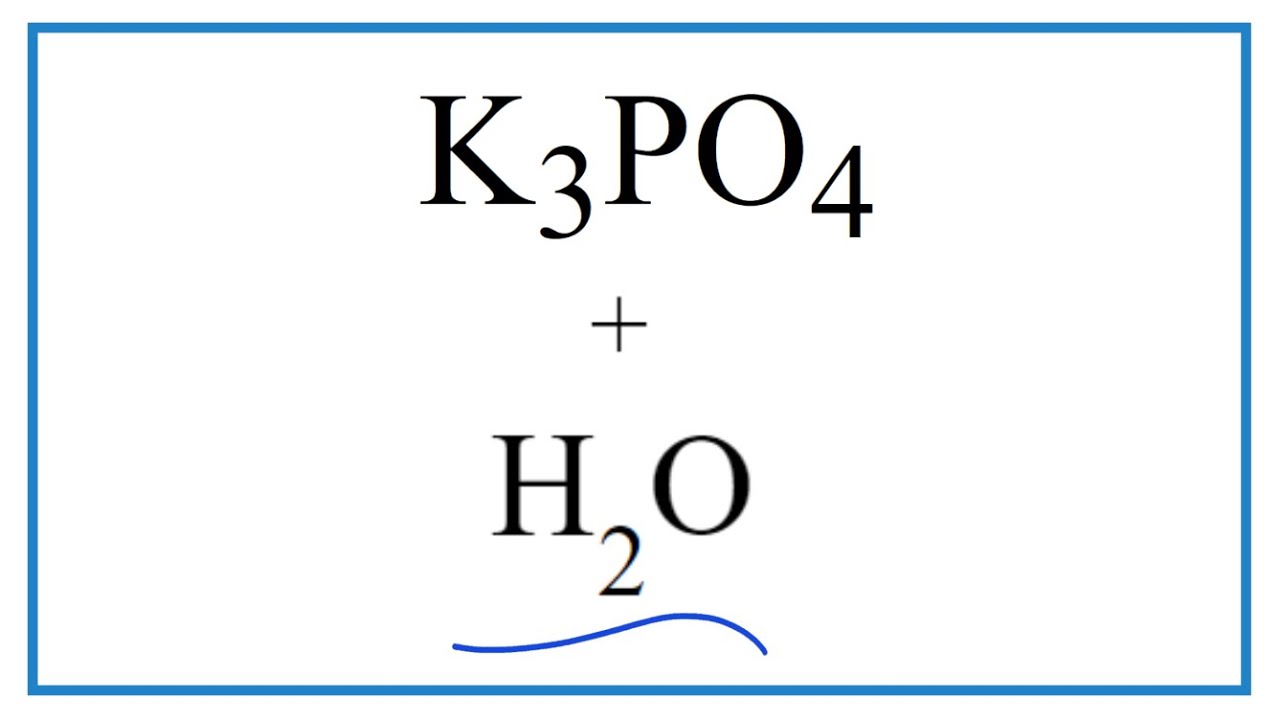
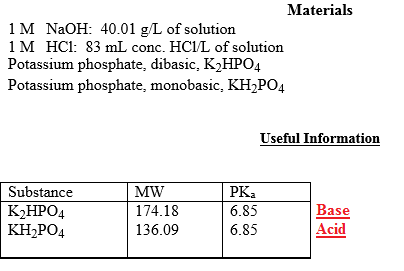
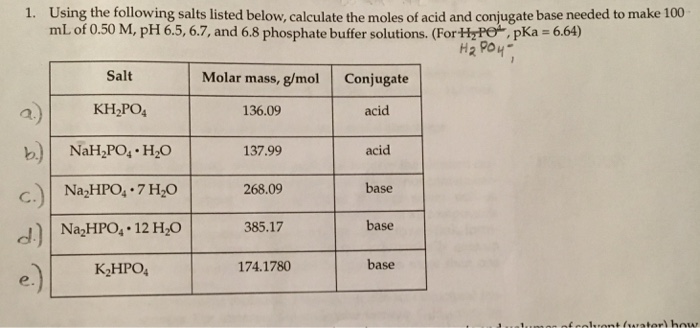
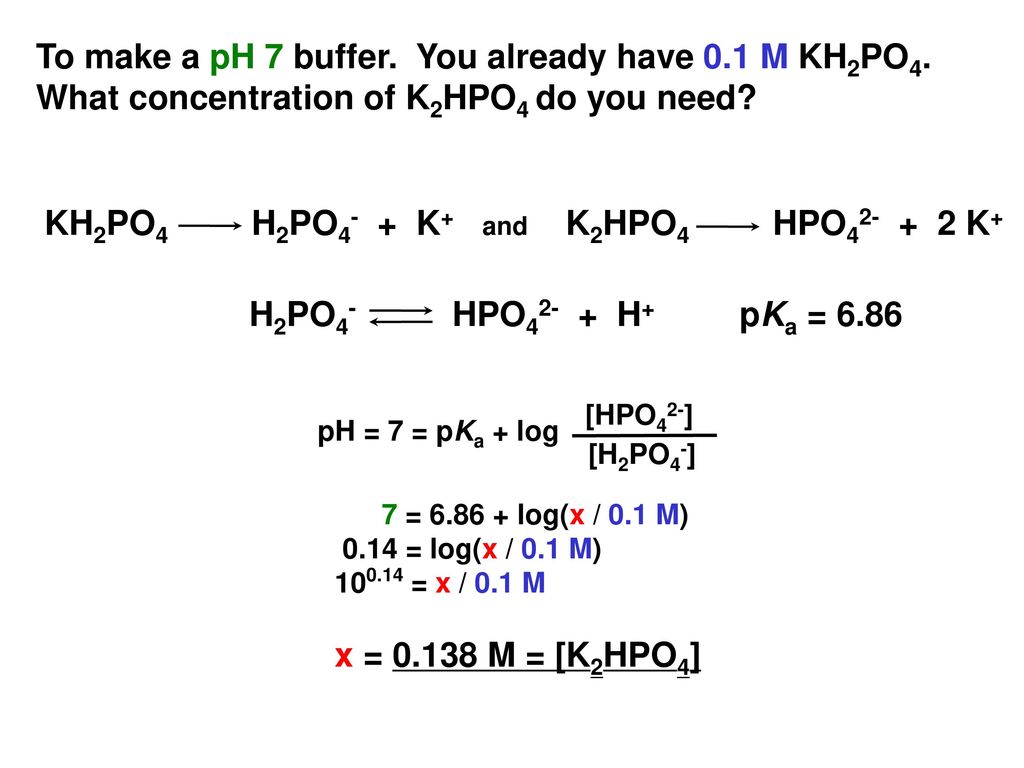
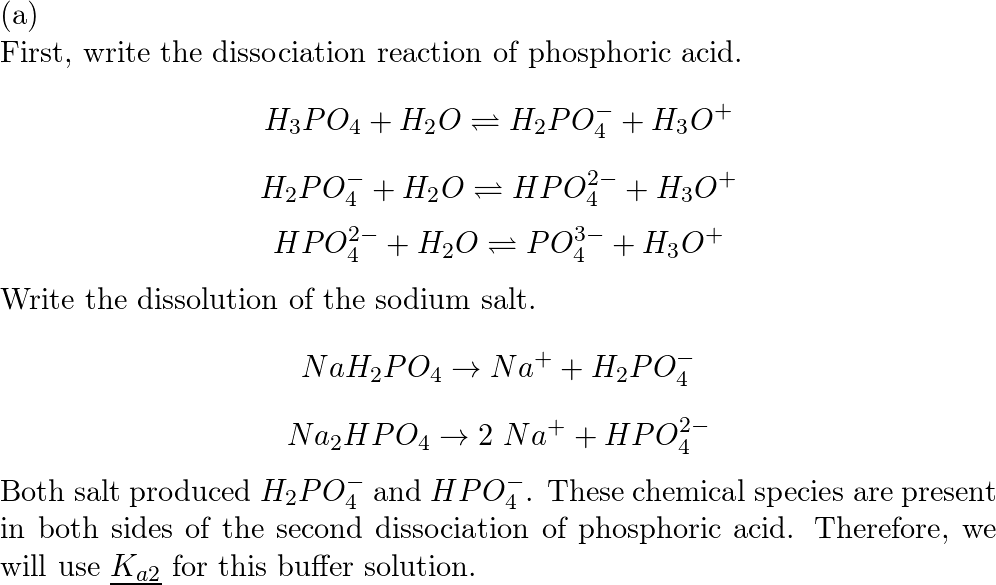Calculate the pH of a buffer solution obtained by dissolving 25.0 g of KH2PO4(s) and 38.0 g of Na2HPO4(s) in water and then diluting to 1.00 L. | Homework.Study.com
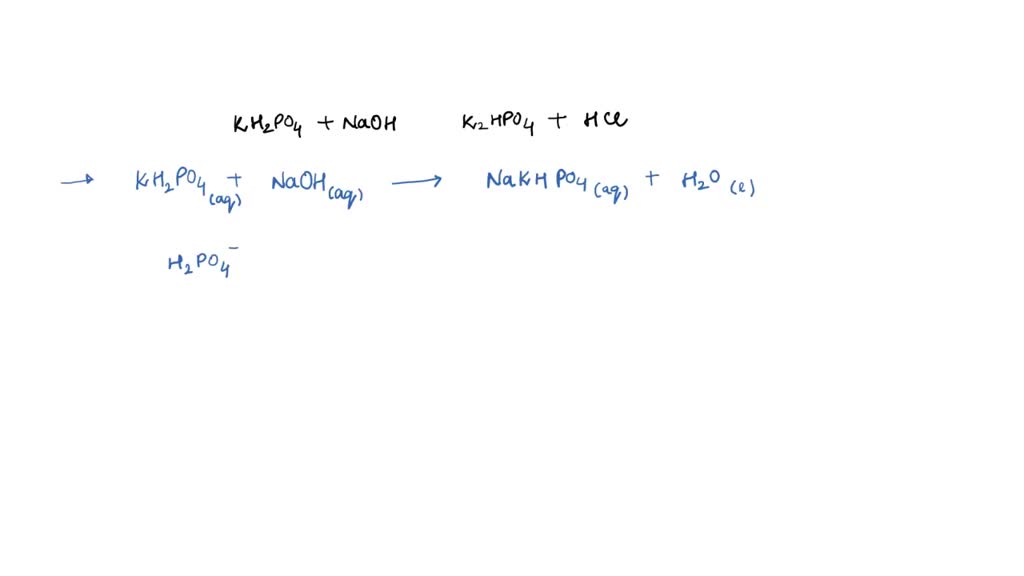
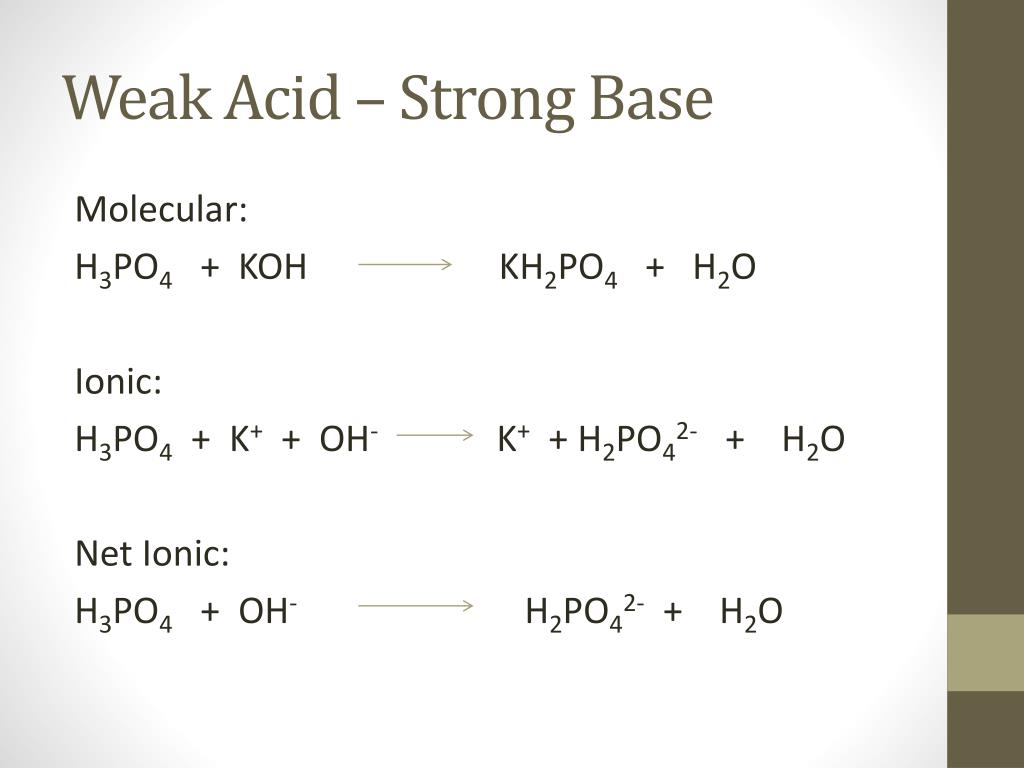
SOLVED: Write the equation and the reaction of the buffer solution KH2PO4 /K2HPO4 when NaOH and HCl is added