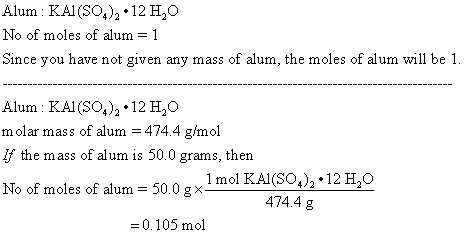PPT - Exp 12 Synthesis: Preparation of Alum Alum: potassium aluminum sulfate dodecahydrate PowerPoint Presentation - ID:5409973

Kal(so4)2 12h2o Potash Alum Potassium Alum Stone - Buy Potassium Alum Stone,Potash Alum Stone,Kal(so4)2 12h2o Potassium Alum Stone Product on Alibaba.com
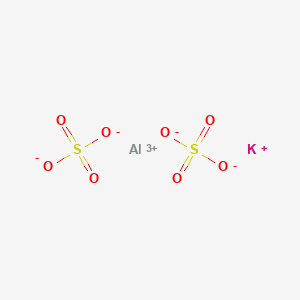
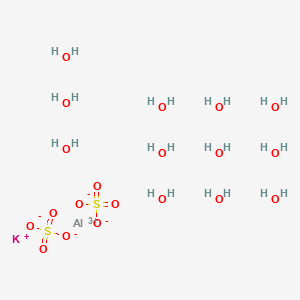
Potash alum is K Al(SO4)2. 12H2O. As a strong eletrolyte, it is considered to be 100% dissociated into K^+, Al^(3+) and SO4^(2-). The solution is acidic because of the hydrolysis of Al^(3+),

Kal(so4)2.12h2o Aluminium Potassium Sulfate Potassium Alum Powder - Buy Potassium Alum Powder,Aluminium Potassium Sulfate Powder,Kal(so4)2 12h2o Potassium Alum Product on Alibaba.com

Potash alum is K Al(SO4)2. 12H2O. As a strong eletrolyte, it is considered to be 100% dissociated into K^+, Al^(3+) and SO4^(2-). The solution is acidic because of the hydrolysis of Al^(3+),
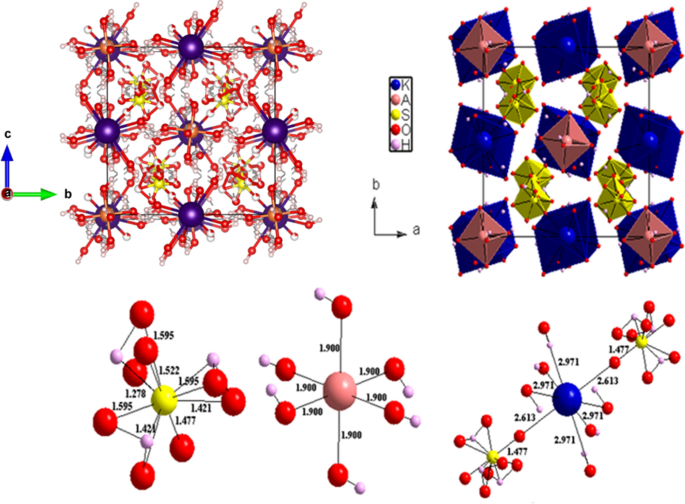
Experimental Investigation and Kinetic Modeling of Potassium Alum Dodecahydrate Thermal Decomposition

Kal(so4)2 12h2o Aluminum Potassium Sulfate Lump Potassium Alum Potash Alum - Buy Potash Alum,Aluminum Potassium Sulfate,Kal(so4)2 12h2o Potash Alum Product on Alibaba.com

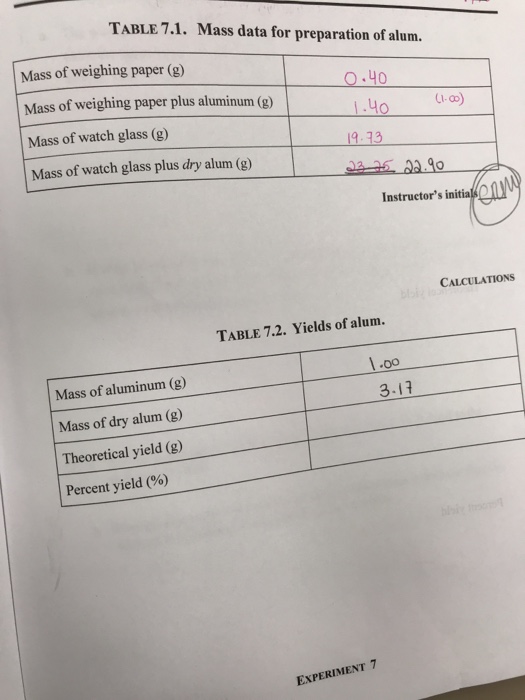
SOLVED: To synthesize alum, KAl(SO4)2 • 12H2O, 1.39 g of solid aluminum are reacted with excess potassium hydroxide and sulfuric acid as described in the experimental section of the writeup in the
![Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S2405844021001638-gr3.jpg)
Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect

Anomalous longitudinal acoustic phonon and elastic constant in potassium alum KAl(SO4)2·12H2O single crystal studied by Brillouin spectroscopy - ScienceDirect

High Quality Potash Alum Kal(so4)2.12h2o - Buy High Purity Aluminium Potassium Sulfate,Aluminium Potassium Sulphate For Food Addictive,Food Grade Aluminium Potassium Sulphate Product on Alibaba.com