Question Video: Calculating the Percentage by Mass of Water in Alum Given Its Chemical Formula | Nagwa

Figure 3 from Vibrational spectra of hexaaquacomplexes. XII. On the possible anion disorder in selenate alums: prediction of the crystal structure and vibrational spectra of KAl(SeO4)2·12H2O and related alums | Semantic Scholar
Types of the coordination polyhedra of K + in K(Al,Cr)(SO 4 ) 2 ⋅12H 2... | Download Scientific Diagram

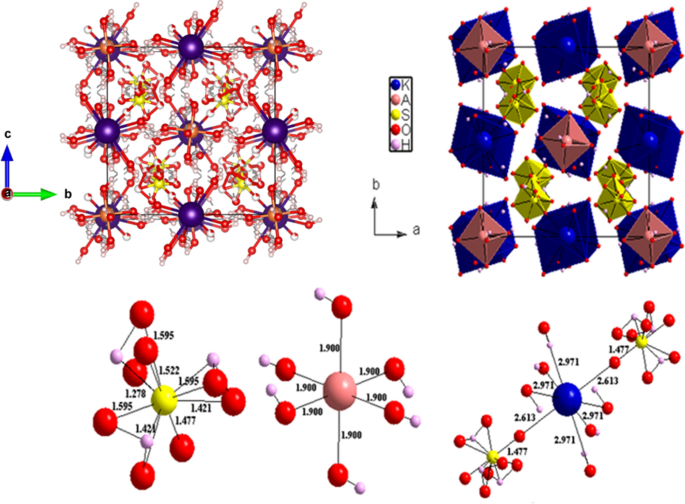
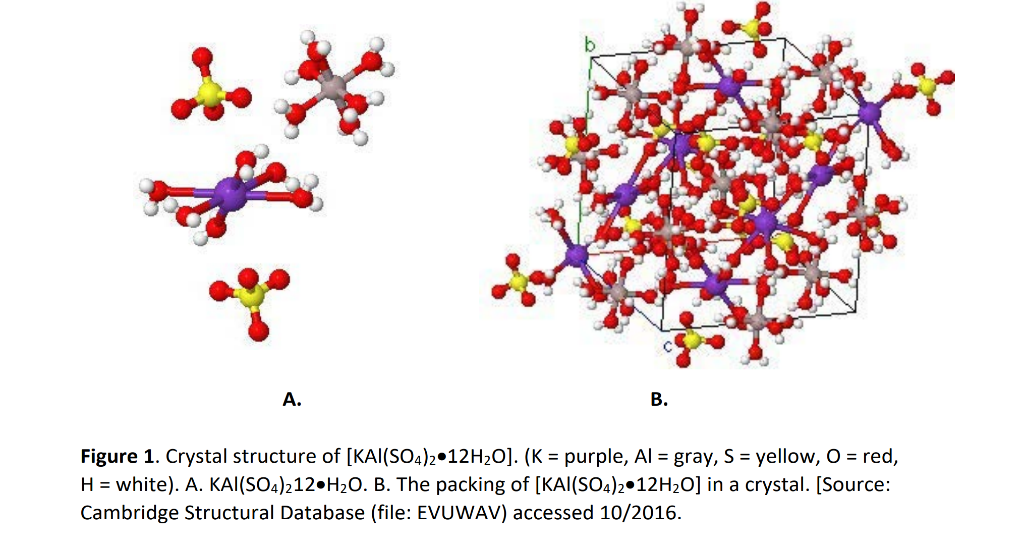
Investigations on KAl(SO4)2∙12H2O: A Candidate α-Alum Material for Energy Storage Applications | SpringerLink

Anomalous longitudinal acoustic phonon and elastic constant in potassium alum KAl(SO4)2·12H2O single crystal studied by Brillouin spectroscopy - ScienceDirect
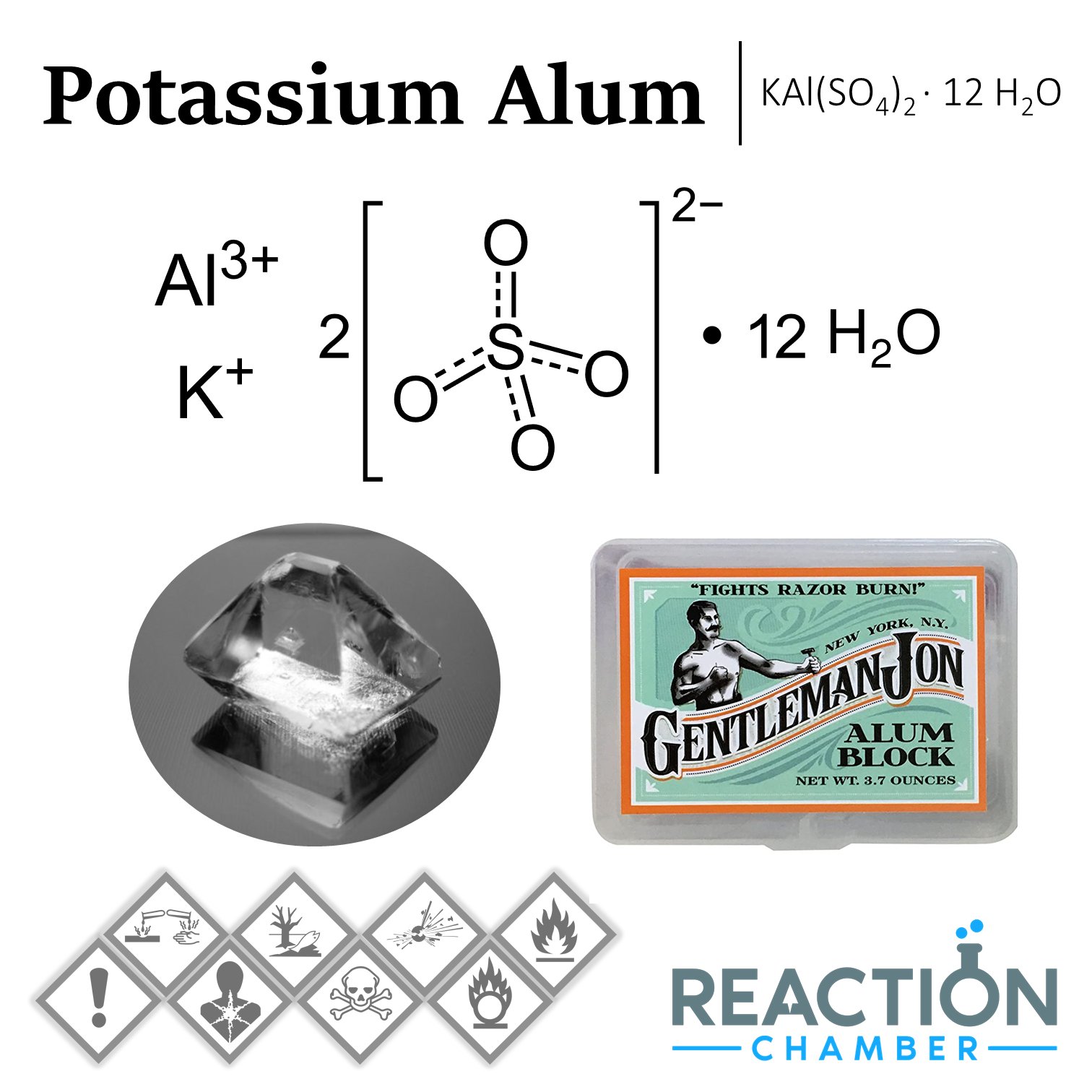
reactionchamber on Twitter: "Potassium alum is the double sulfate salt of potassium and aluminium. It is commonly encountered as the dodecahydrate, KAl(SO4)2·12H2O. It crystallizes in an octahedral structure in neutral solution. #alum #
![Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S2405844021001638-sc1.jpg)
Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect
Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene
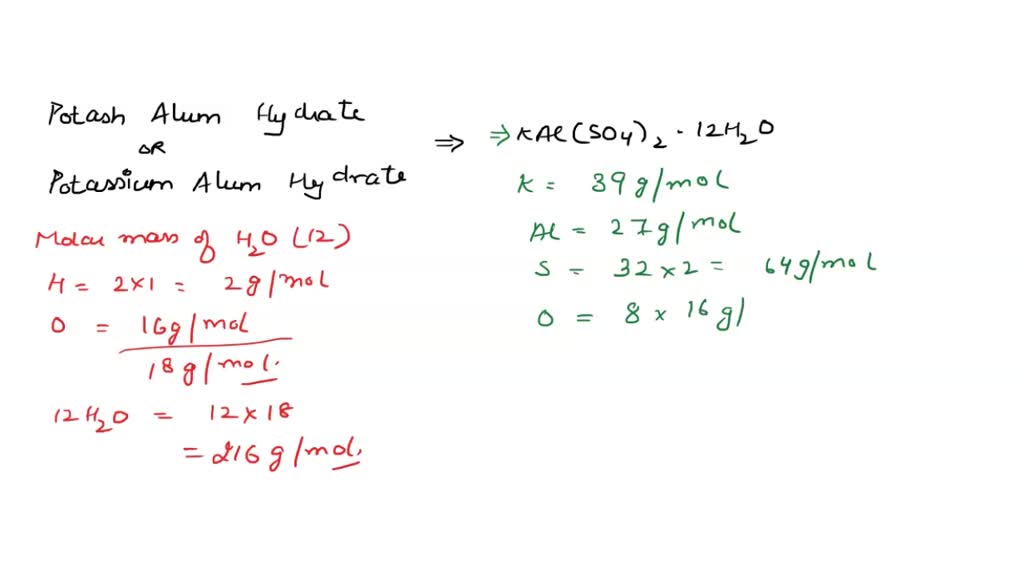
SOLVED: What is the mass percent of water in the Potassium Alum Hydrate: KAl (SO4)2·12H2O? A) 8.33% B) 91.67% C) 21.62% D) 54.43% E) 45.57%