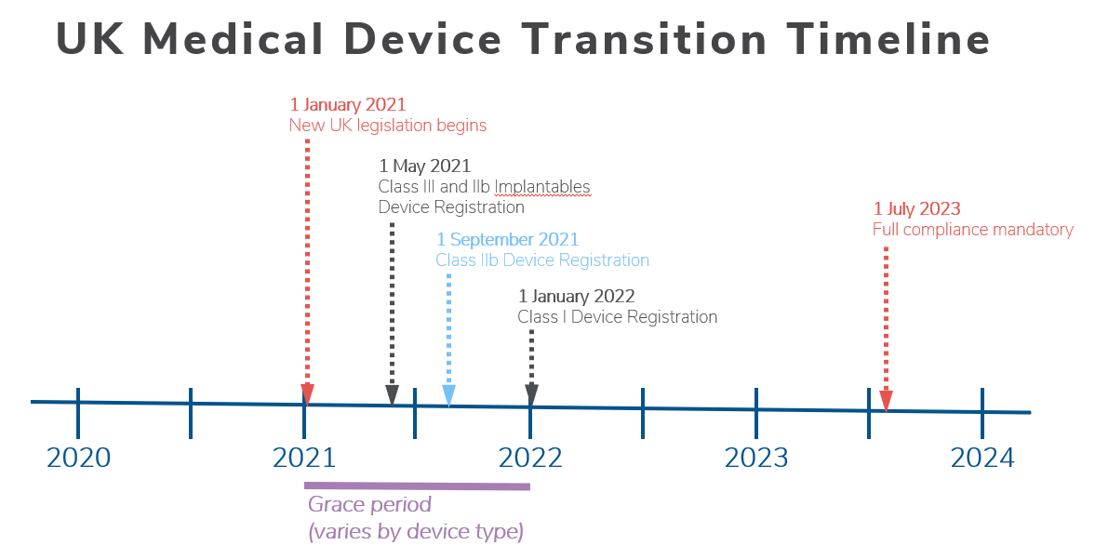
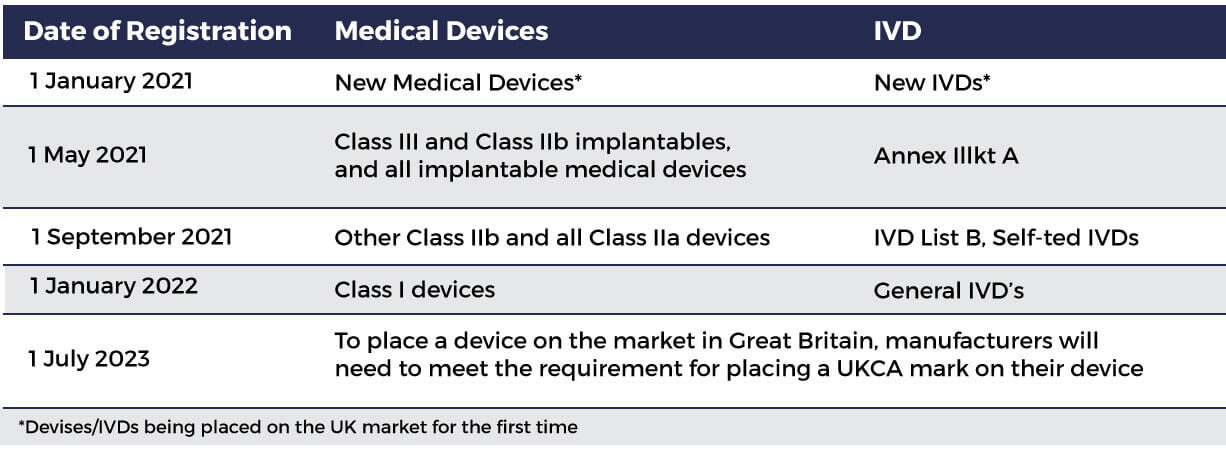
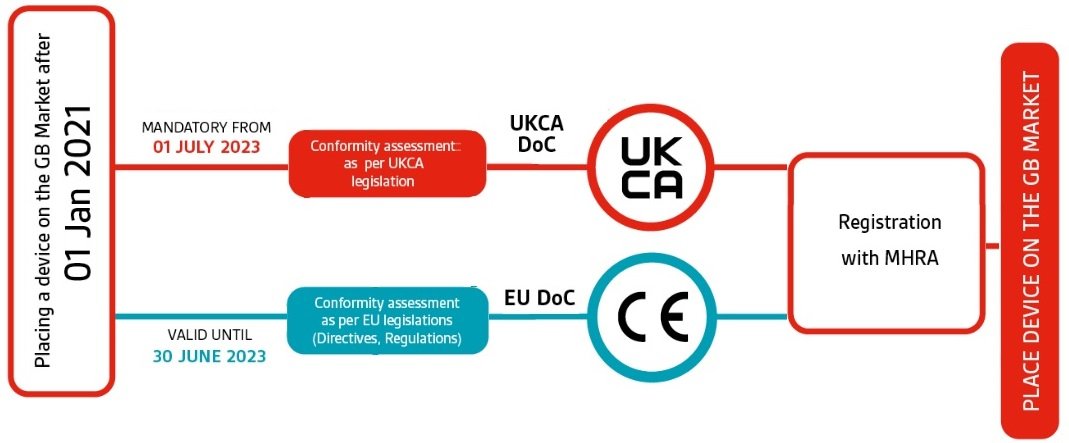
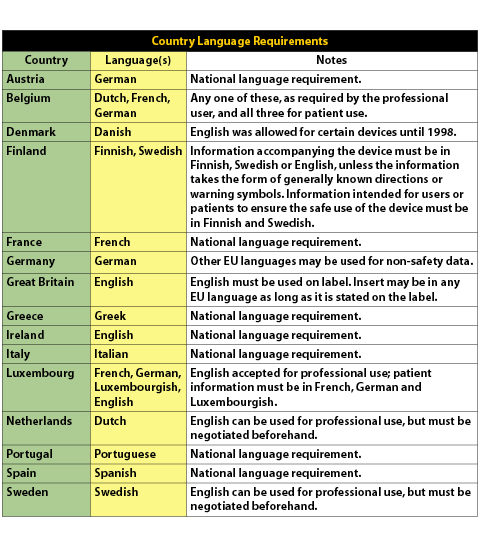
New UK medical device regulation spells potential trial concerns for some IVD players - Medical Device Network

MHRA guidance on registration and deadlines for medical devices and IVDs in Great Britain and Northern Ireland

A Strategic Technology Roadmap for the UK In Vitro Diagnostics Industry – Cambridge Design Partnership
Top 10 Questions regarding the UK Responsible Person and medical device/IVD registration with the MHRA

Top 10 Questions regarding the UK Responsible Person and medical device/IVD registration with the MHRA

IVD Directive to IVD Regulation (EU 2017/746) Transition – 8 Months Remaining - Voisin Consulting Life Sciences