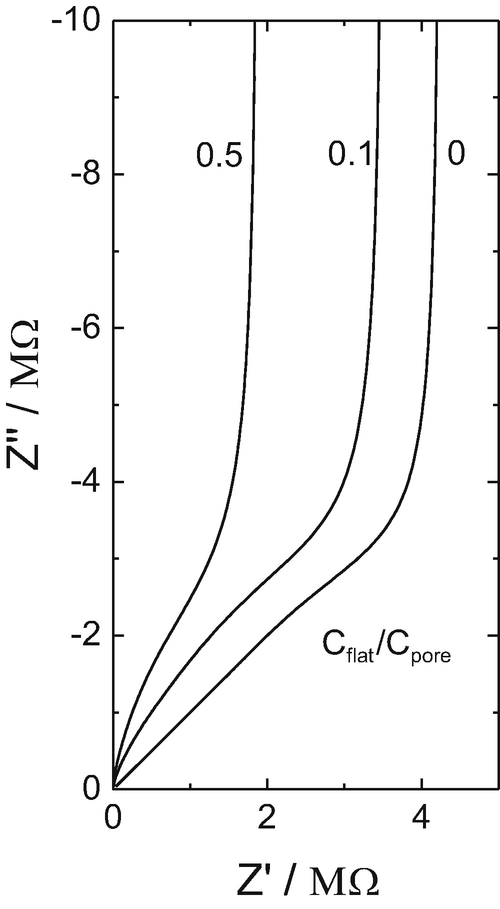
Study of the electrolyte-insulator-semiconductor field-effect transistor (EISFET) with applications in biosensor design - ScienceDirect
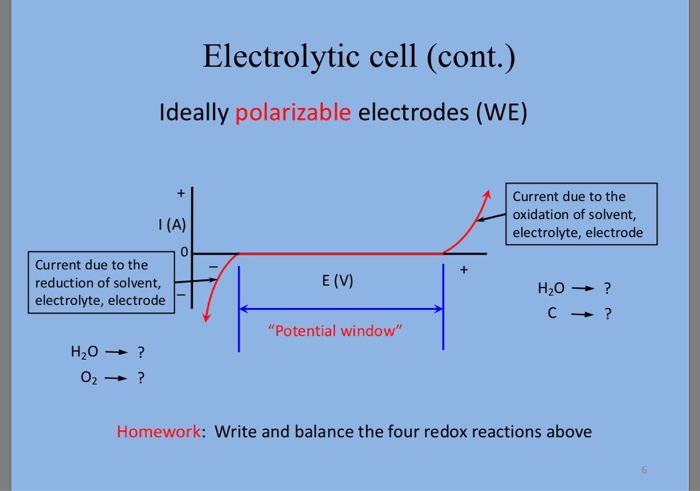
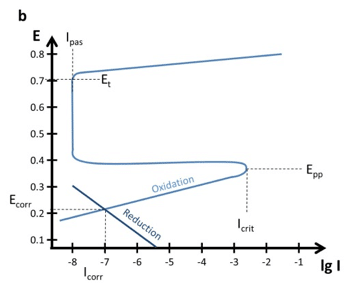
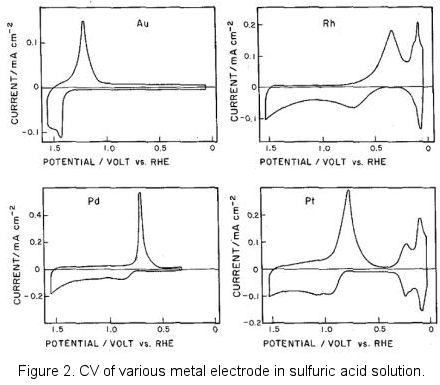
CHM 432: ADVANCED ELECTROCHEMISTRY CONTENTS 1.0. ELECTRICAL DOUBLE LAYER • Faradaic and Non faradaic current • Mass Transfe

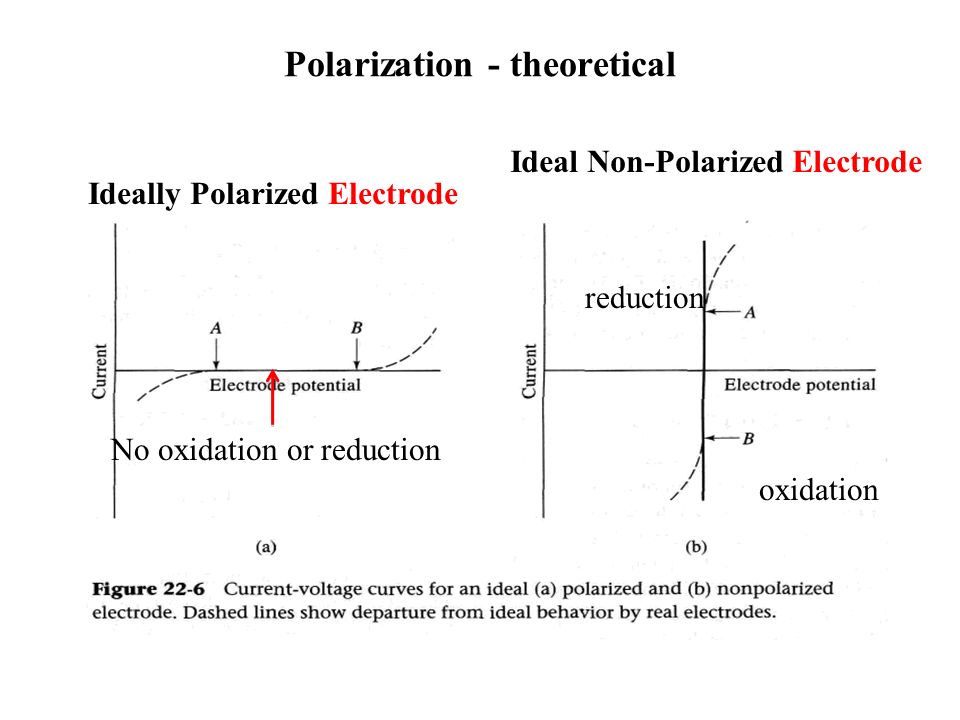
Difference Between Polarizable and Non Polarizable Electrode | Compare the Difference Between Similar Terms

SOLVED: 47 00/0 Question 9/18 (1 p ) Answer is mandatory In an electrochemical cell, if the potential of one of the electrodes was changing even when the current is kept constant (
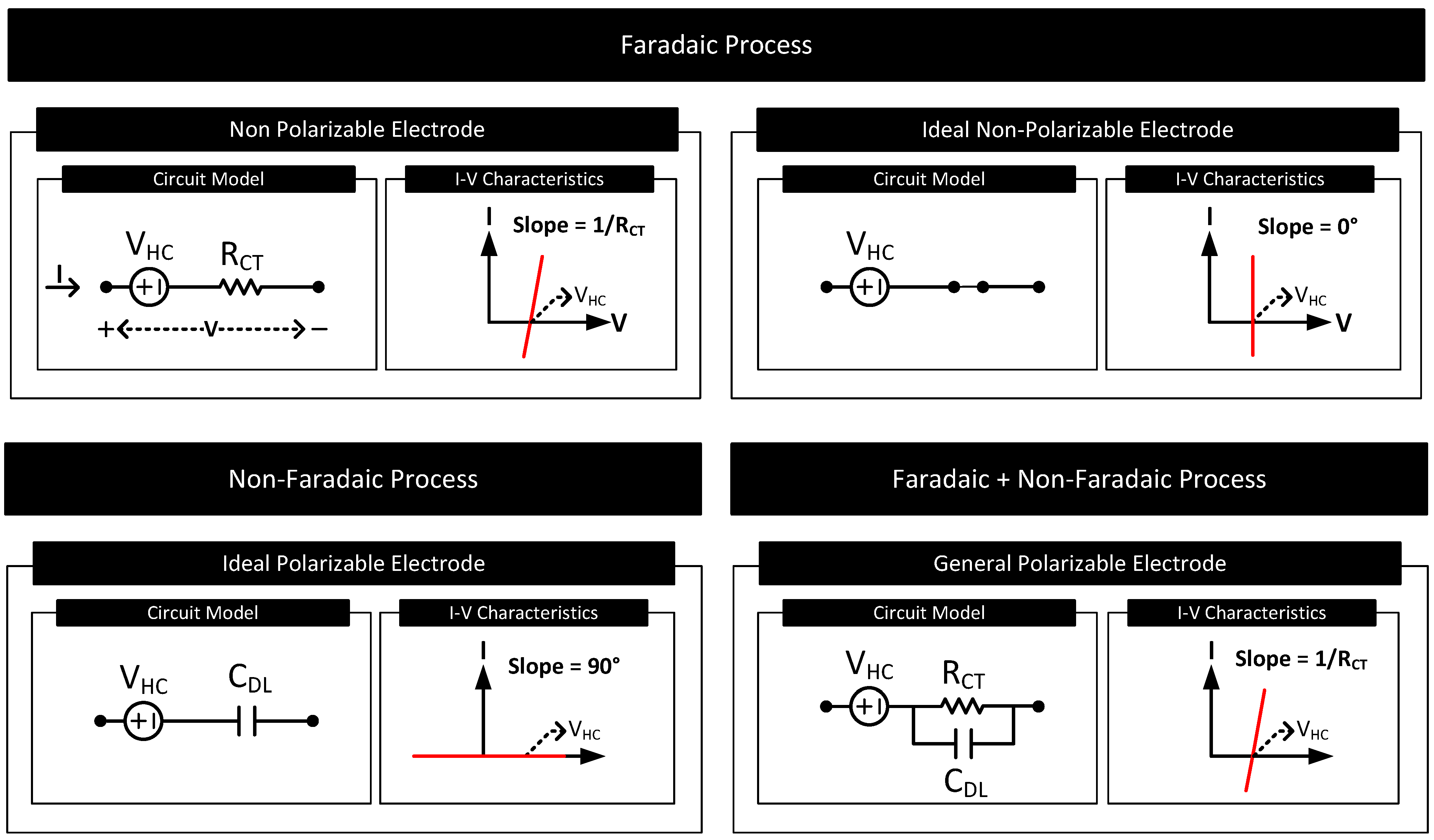
Nanomaterials | Free Full-Text | Human Body–Electrode Interfaces for Wide-Frequency Sensing and Communication: A Review
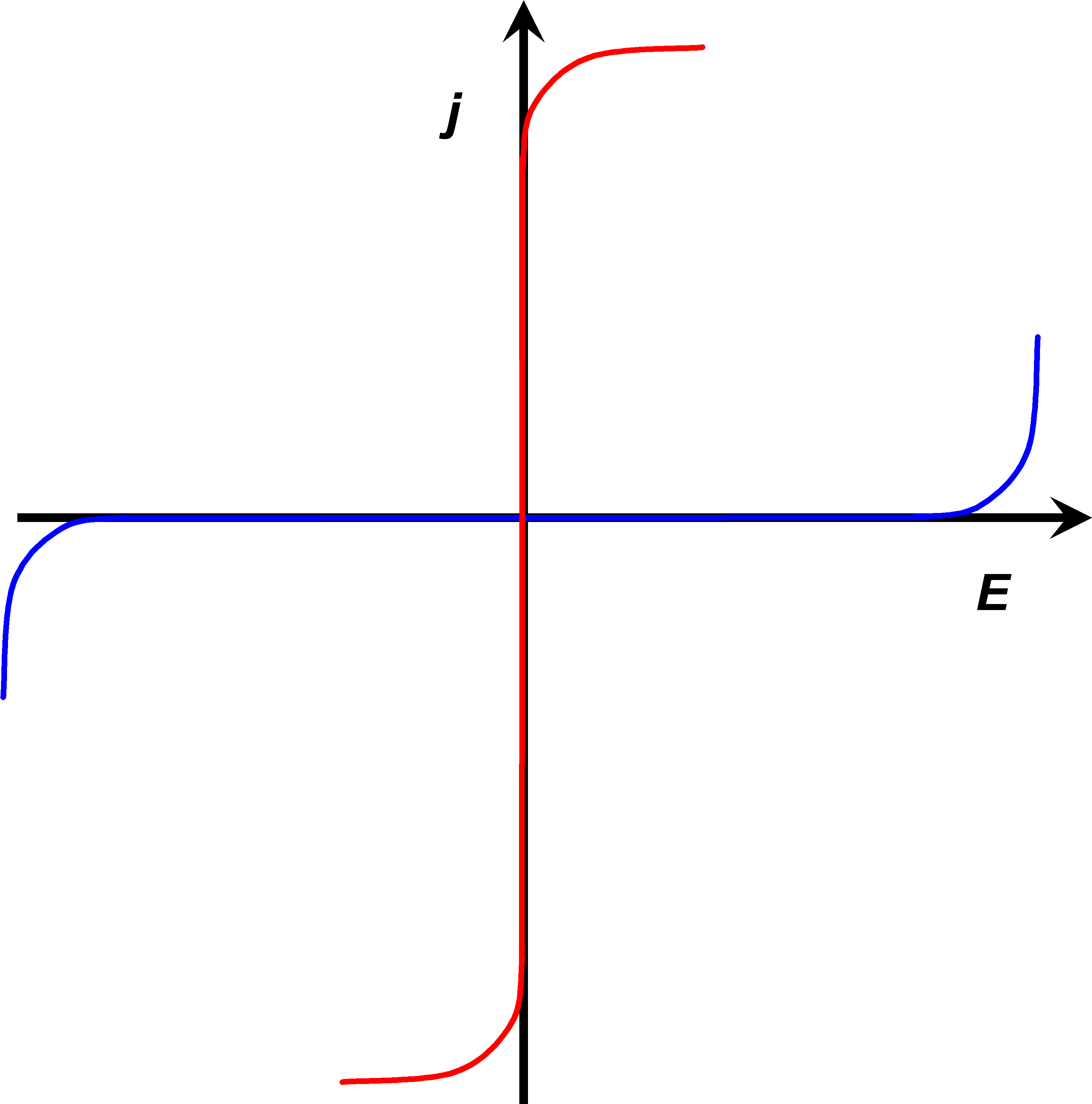
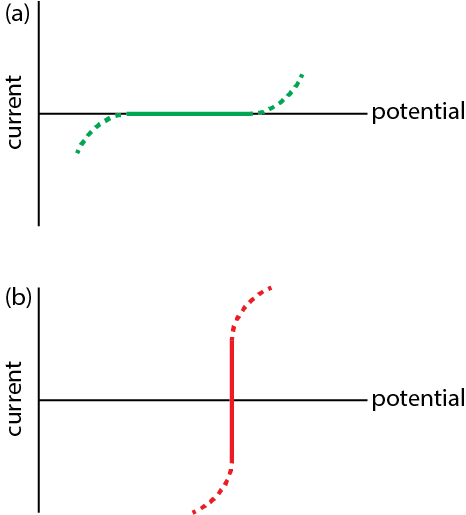
Download Current Voltage Curves Of Nearly Ideally Non Polarizable - Electrode PNG Image with No Background - PNGkey.com
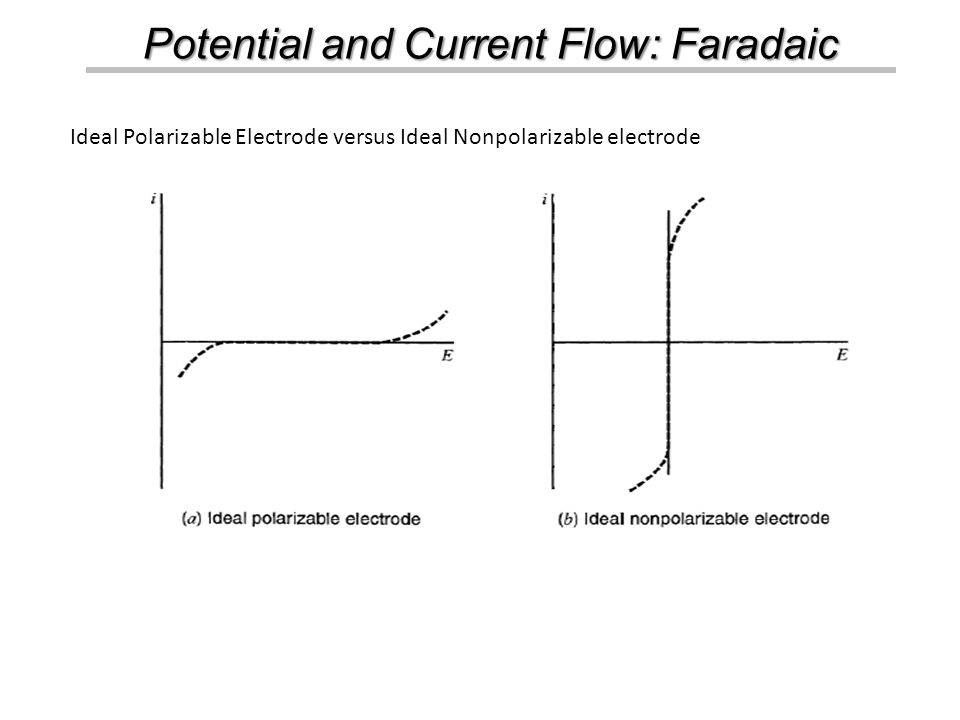
America's Smartest City by Movoto Blog Rated Most Livable City by Places Rated Almanac and The Economist Named among Best in the World Places to Visit. - ppt download

A general thermodynamic theory of the ideal polarized electrode - Transactions of the Faraday Society (RSC Publishing)
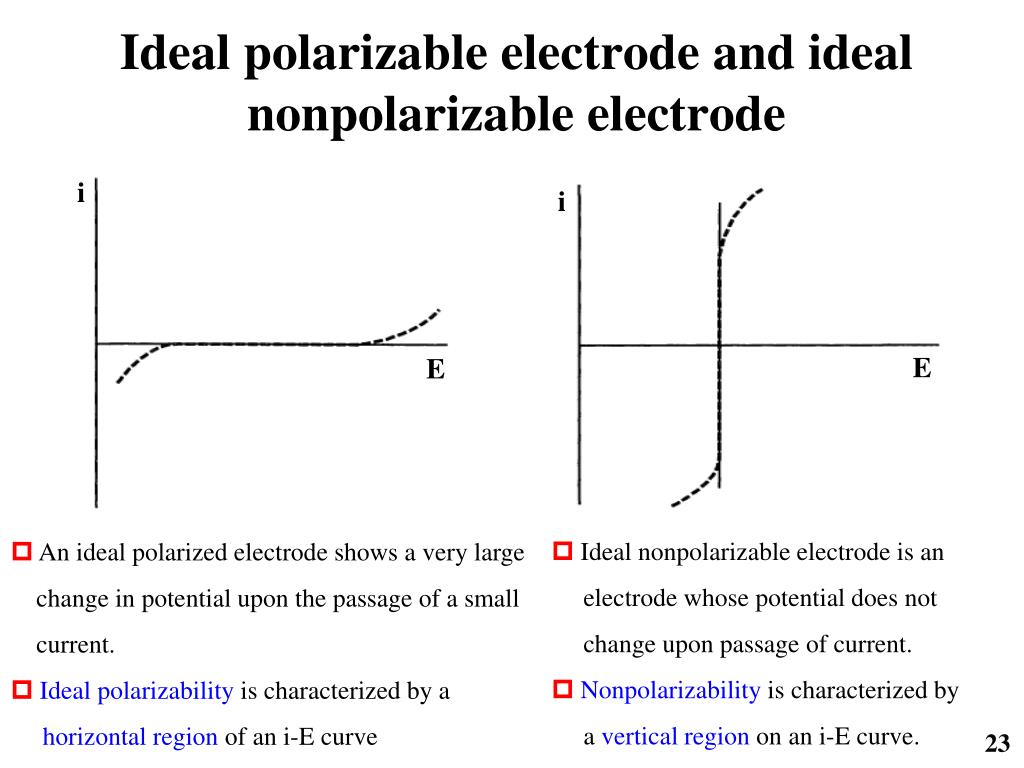
PPT - Electro-Optical Materials Laboratory Department of Chemical Engineering PowerPoint Presentation - ID:3931155