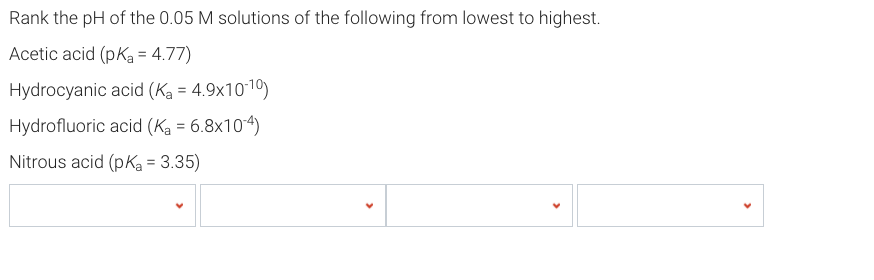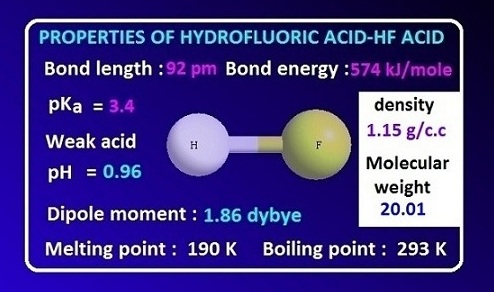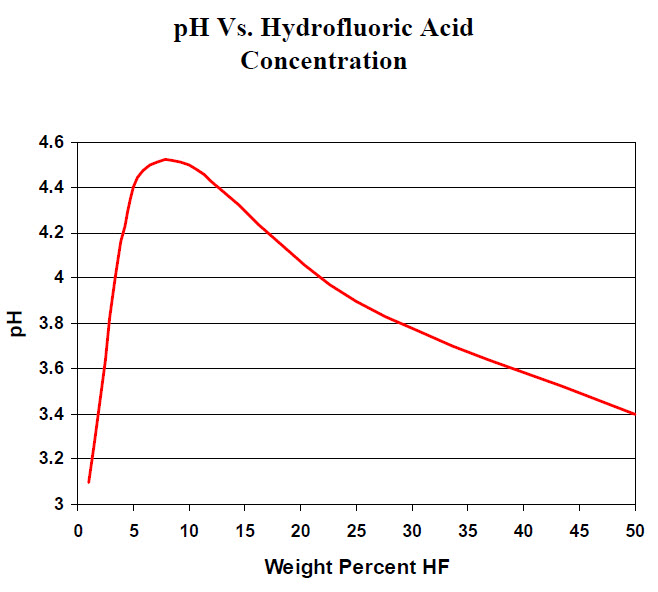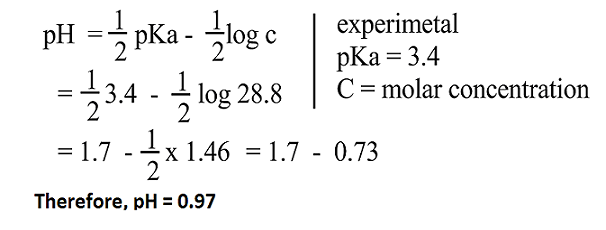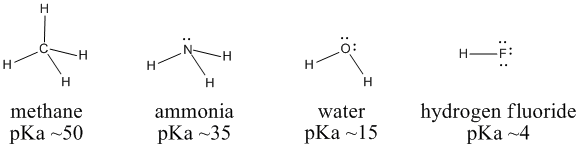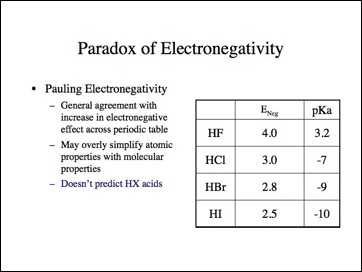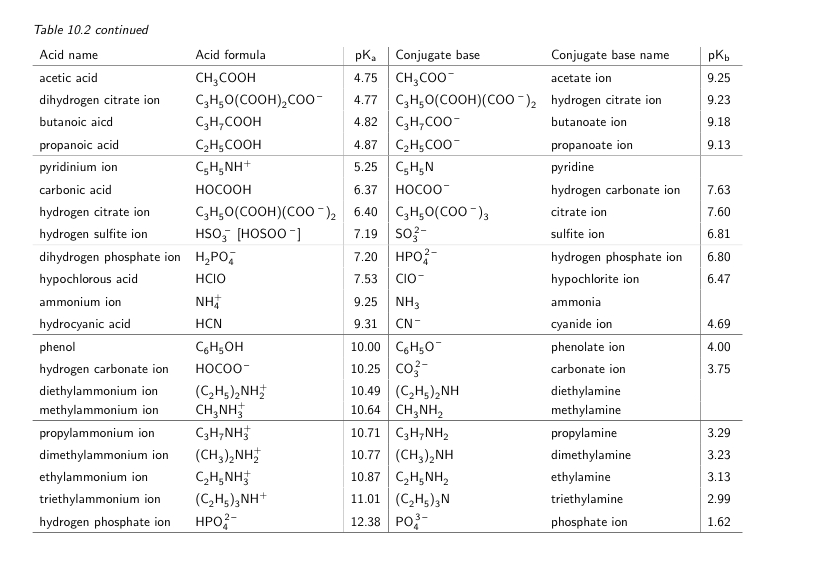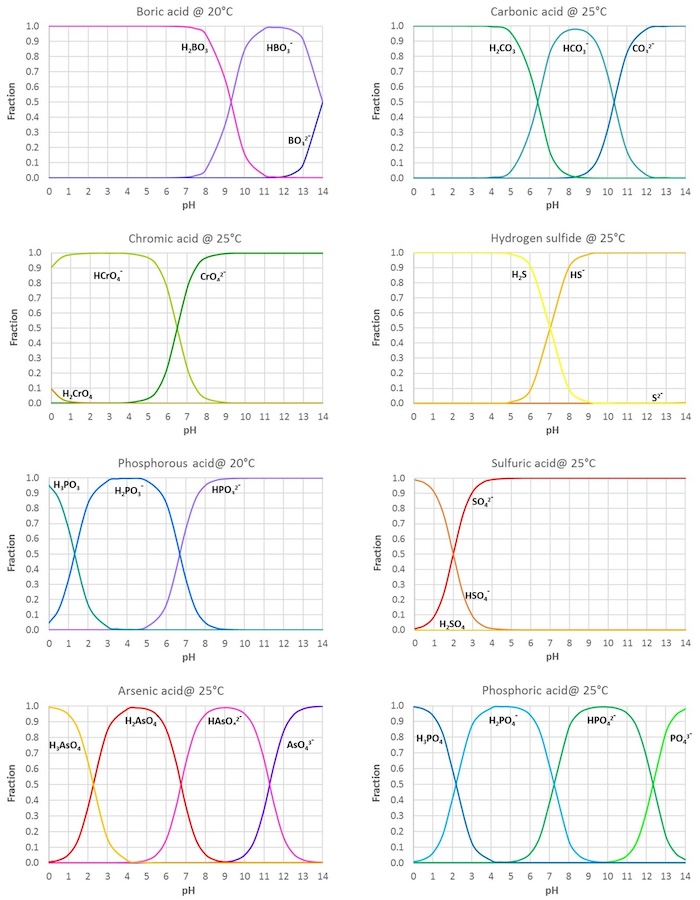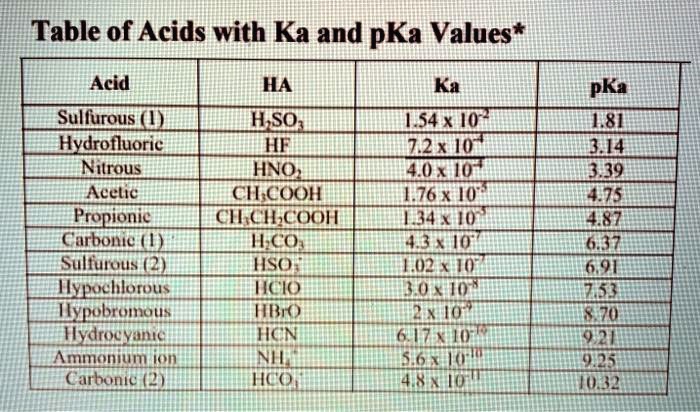
SOLVED: Table of Acids with Ka and pKa Values* Acid HA Ka Sullurous ( 4) HSO; LS4X Ho? Hydrofluoric HF Z2X0I Mutrous HNO; 40Xl0i Aeelic CICOOH L7ox I0 Prepionie CLCLCOOH LL4O Cubonie ()

SOLVED: Consider the following unfinished table of weak acids with their corresponding pKa values and Ka values. Weak Acid HNO2 HF pKa 335 6.8 * 10 + HCN 9.31 Arrange these three
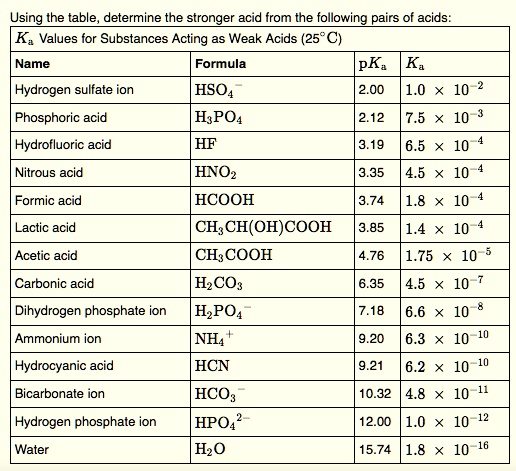

![The pKa values for various precipitants [17]. | Download Scientific Diagram The pKa values for various precipitants [17]. | Download Scientific Diagram](https://www.researchgate.net/publication/339359335/figure/tbl1/AS:860297669640196@1582122356901/The-pKa-values-for-various-precipitants-17.png)
![The pKa values for various precipitants [17]. | Download Scientific Diagram The pKa values for various precipitants [17]. | Download Scientific Diagram](https://www.researchgate.net/publication/339359335/figure/tbl1/AS:860297669640196@1582122356901/The-pKa-values-for-various-precipitants-17_Q320.jpg)