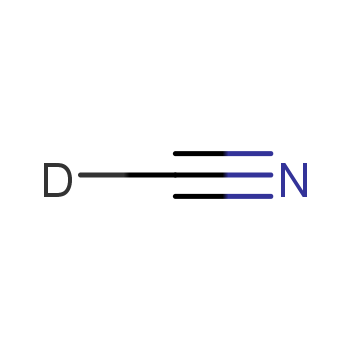
Hydrocyanic Acid Handwritten Chemical Formula Illustration Stock Illustration 1707418036 | Shutterstock

How many grams of solid potassium cyanide should be added to 1.00 L of 0.240 M hydrocyanic acid solution to prepare a buffer with a pH of 10.100? (Ka for hydrocyanic acid =

SOLVED: for hydrocyanic acid, HCN, is 4.00x10-10. K for benzoic acid, CHsCOOH, is 6.30x10-5. for acetic acid, CH;COOH, is 1.80x10-5 What is the formula for the strongest acid?

Hydrocyanic acid is a notorious poison. In very dilute solutions, however, it does have medical uses. - SuperStock
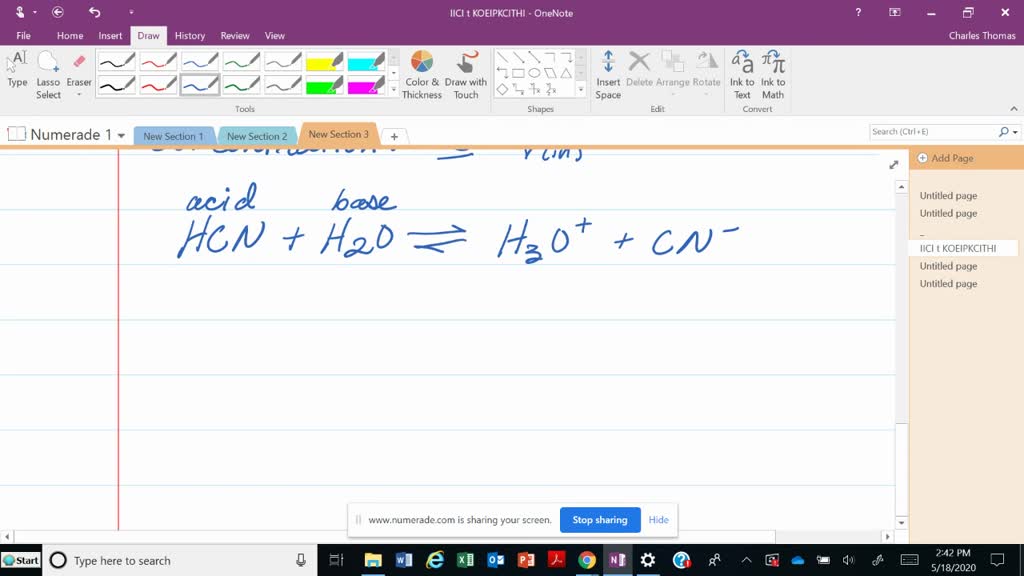
SOLVED:Write an equation for the reaction between hydrocyanic acid, HCN, and water. Label the acid, base, conjugate acid, and conjugate base.
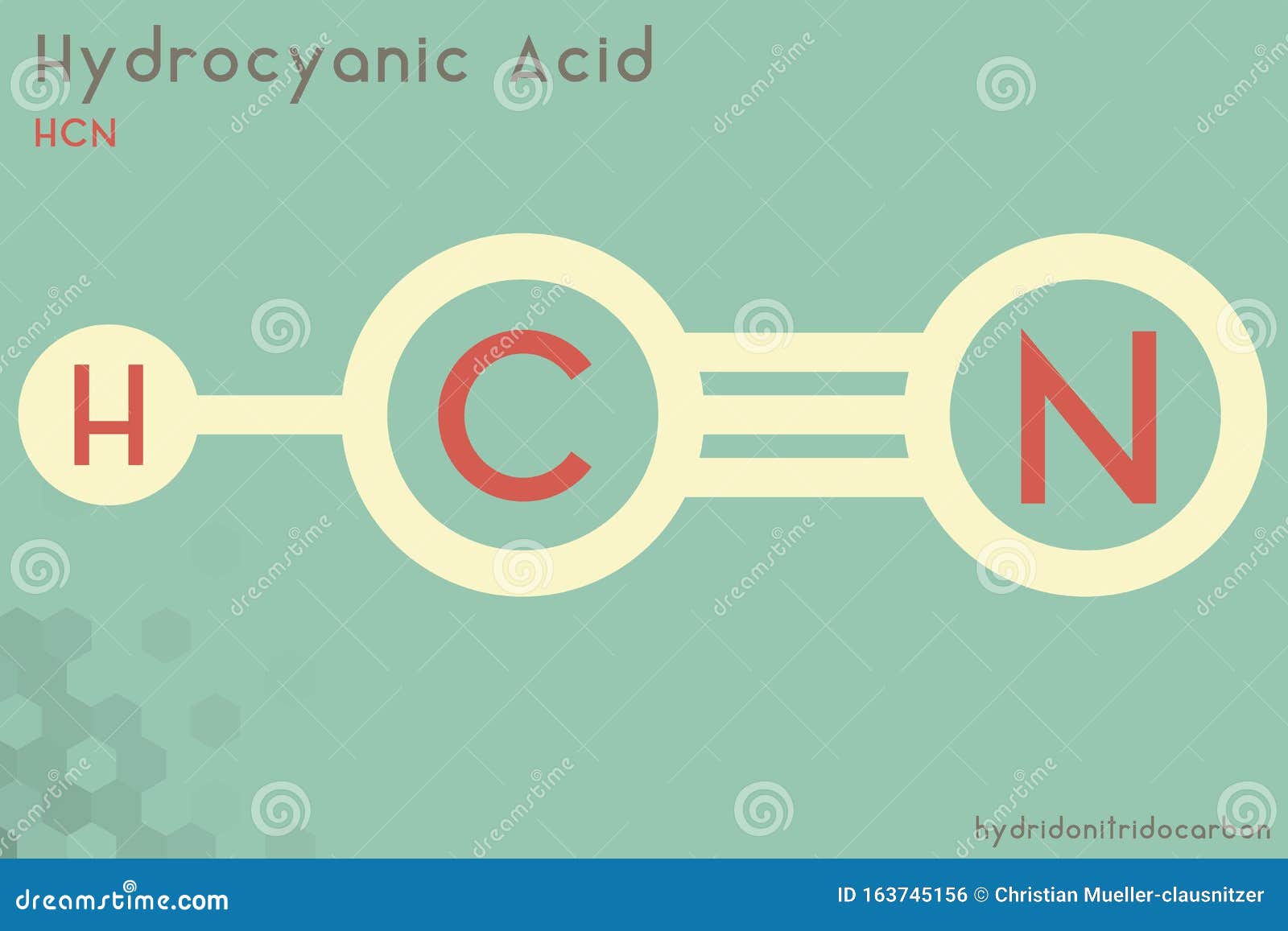
Hydrocyanic Stock Illustrations – 59 Hydrocyanic Stock Illustrations, Vectors & Clipart - Dreamstime

Hydrocyanic Stock Illustrations – 59 Hydrocyanic Stock Illustrations, Vectors & Clipart - Dreamstime
10: Chemical reactions leading to formation of hydrocyanic acid (HCN)... | Download Scientific Diagram
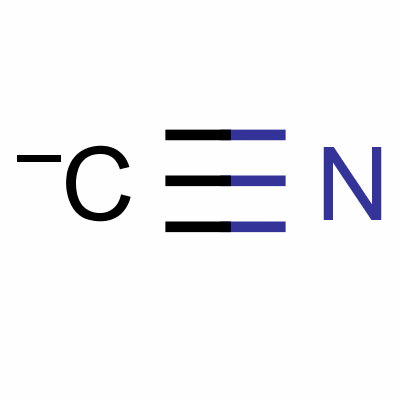
Hydrocyanic acid 57-12-5, China Hydrocyanic acid 57-12-5 Manufacturers, China Hydrocyanic acid 57-12-5 Suppliers