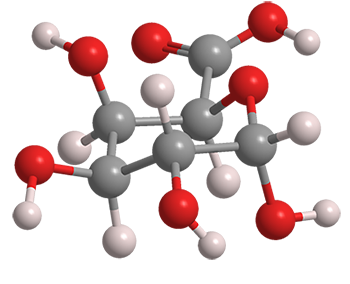
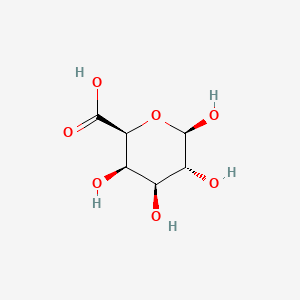
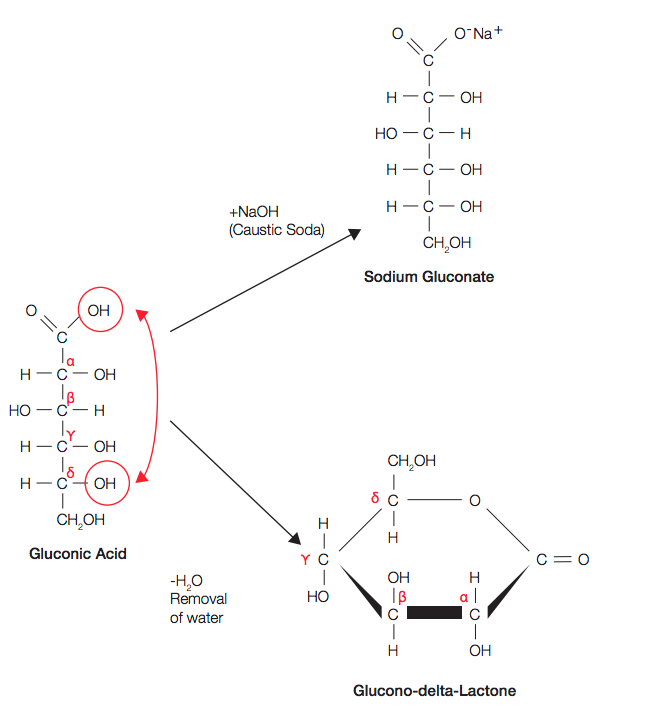
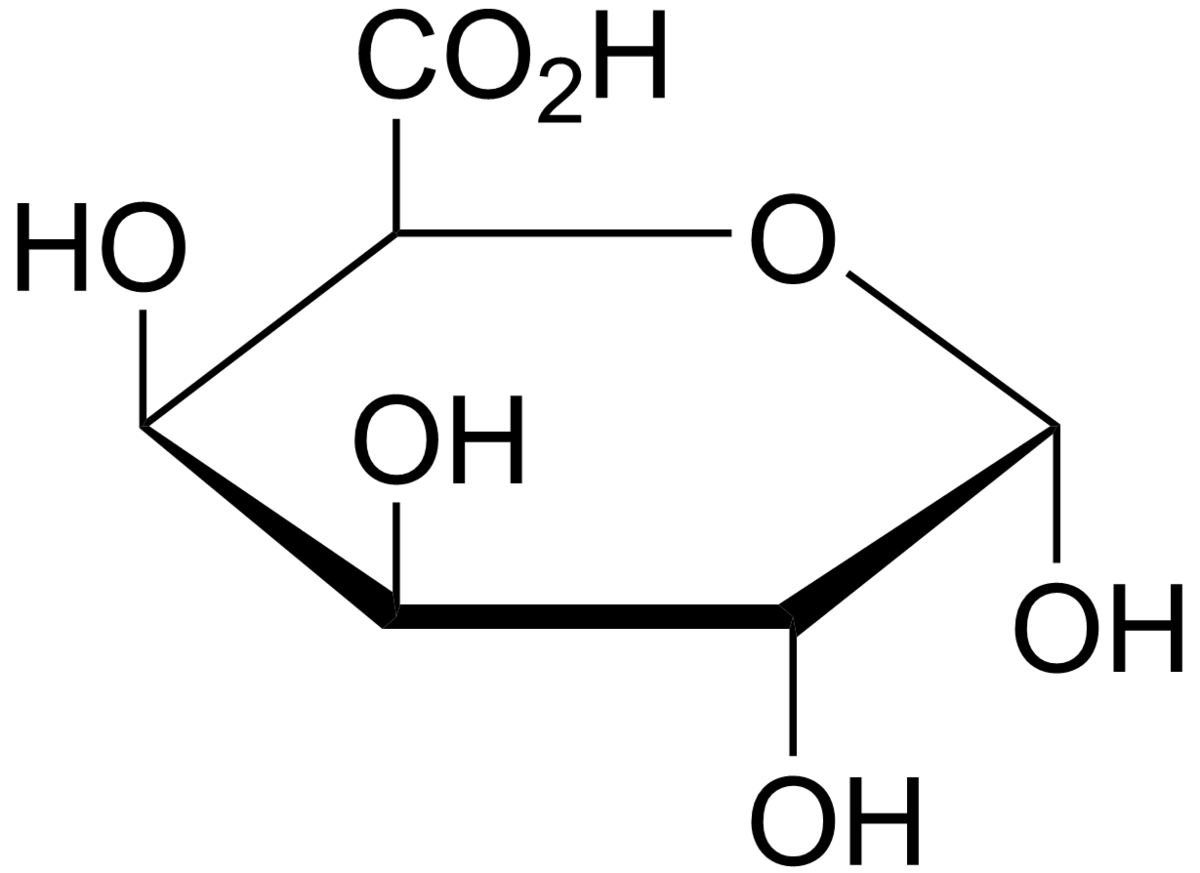
Determination of the pKa of glucuronic acid and the carboxy groups of heparin by 13C-nuclear-magnetic-resonance spectroscopy. - Abstract - Europe PMC

Simultaneous fermentation of galacturonic acid and five-carbon sugars by engineered Saccharomyces cerevisiae - ScienceDirect
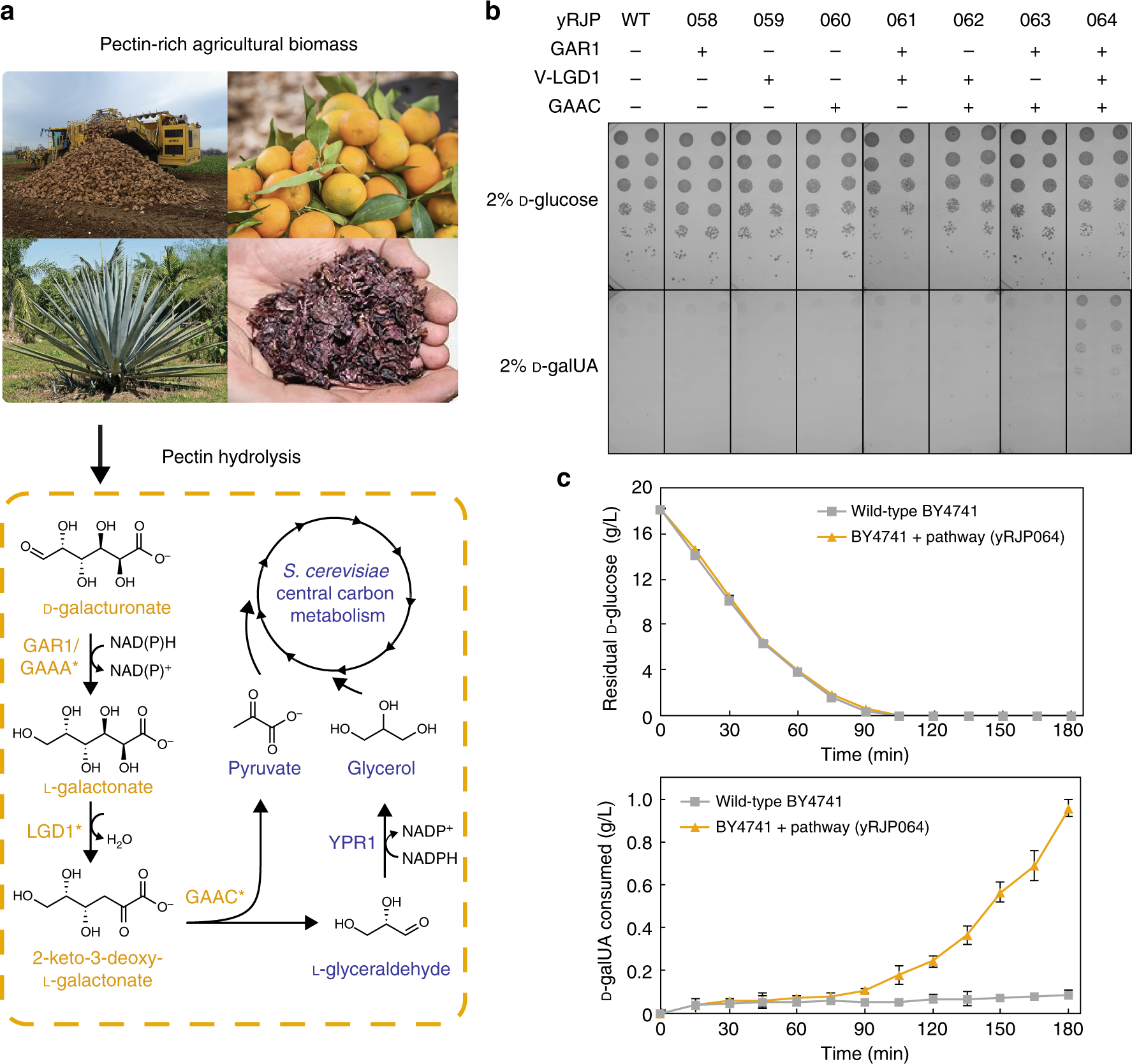
Engineering Saccharomyces cerevisiae for co-utilization of d-galacturonic acid and d-glucose from citrus peel waste | Nature Communications

d-Galacturonic Acid: A Highly Reactive Compound in Nonenzymatic Browning. 2. Formation of Amino-Specific Degradation Products | Journal of Agricultural and Food Chemistry

5894-59-7 | Digalacturonic Acid (>85%) | D-4-O-α-D-Galactopyranuronosyl-galacturonic Acid; | C12H18O13 | TRC
Improvement of nanofibrillation efficiency of α-chitin in water by selecting acid used for surface cationisation - RSC Advances (RSC Publishing) DOI:10.1039/C2RA22271J

Structure of galacturonic acid (a), glucuronic acid (b) galacturonic... | Download Scientific Diagram

Engineering Filamentous Fungi for Conversion of d-Galacturonic Acid to l-Galactonic Acid | Applied and Environmental Microbiology
![PDF] Determination of the pKa of glucuronic acid and the carboxy groups of heparin by 13C-nuclear-magnetic-resonance spectroscopy. | Semantic Scholar PDF] Determination of the pKa of glucuronic acid and the carboxy groups of heparin by 13C-nuclear-magnetic-resonance spectroscopy. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a4675c65fa0813e16ad330bbabe94f30cb0dd96d/3-Figure1-1.png)
PDF] Determination of the pKa of glucuronic acid and the carboxy groups of heparin by 13C-nuclear-magnetic-resonance spectroscopy. | Semantic Scholar

Influence of the Carboxylic Function on the Degradation of d-Galacturonic Acid and Its Polymers | Journal of Agricultural and Food Chemistry

d-Galacturonic Acid: A Highly Reactive Compound in Nonenzymatic Browning. 2. Formation of Amino-Specific Degradation Products | Journal of Agricultural and Food Chemistry

d-Galacturonic Acid: A Highly Reactive Compound in Nonenzymatic Browning. 2. Formation of Amino-Specific Degradation Products | Journal of Agricultural and Food Chemistry