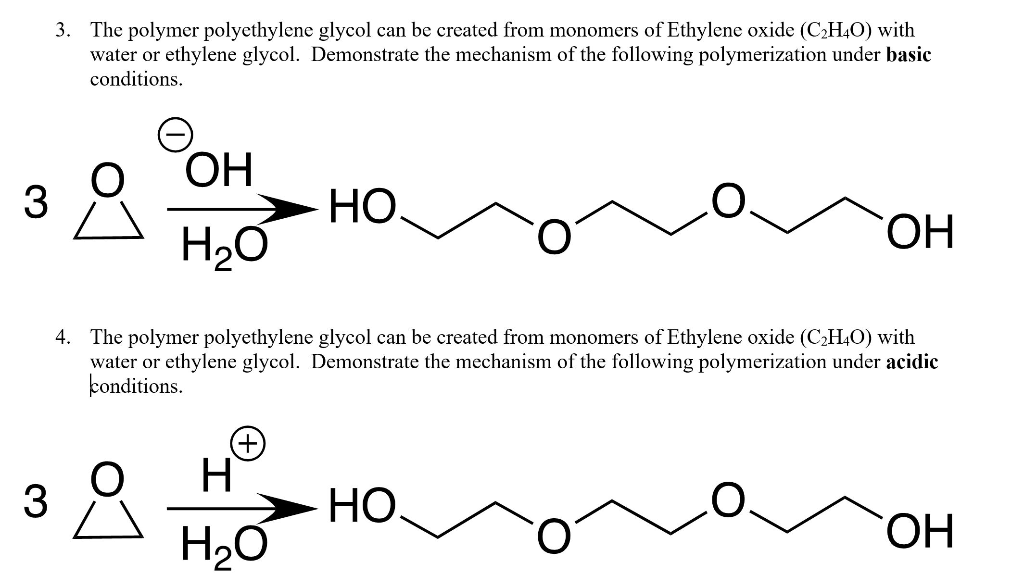
Polymerization of Ethylene Oxide, Propylene Oxide, and Other Alkylene Oxides: Synthesis, Novel Polymer Architectures, and Bioconjugation | Chemical Reviews

Synthesize the following compound from ethylene oxide and any other reagents of your choice. | Homework.Study.com
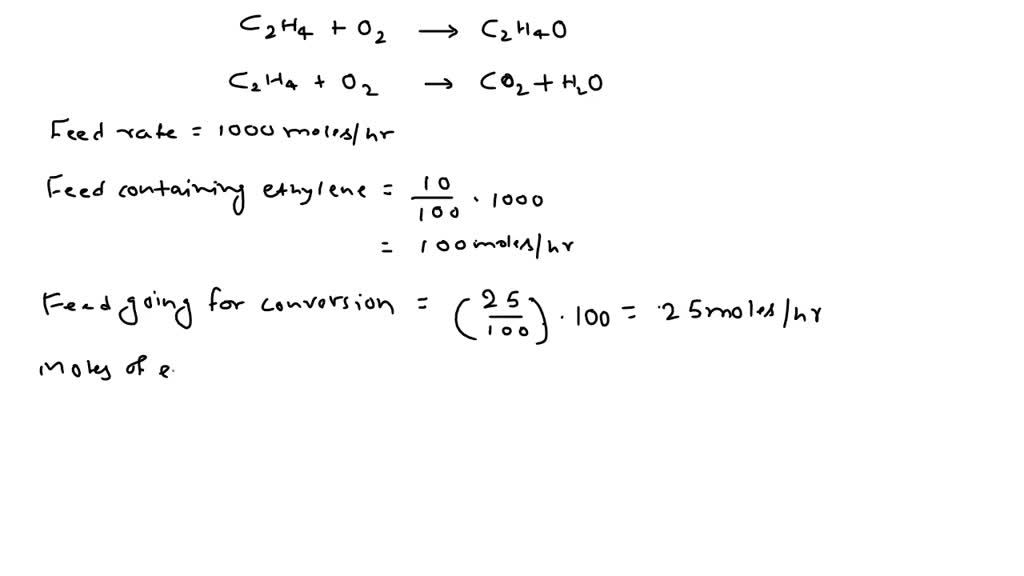
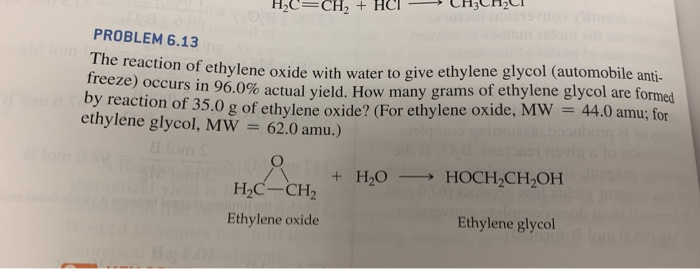
SOLVED: Ethylene glycol (HOCH, CH, OH), used as an antifreeze, is produced by reacting ethylene oxide with water. A side reaction produces an unwanted dimer, DEG: C2H40 + H2O → HOCH2CH2OH HOCH2CH2OH +
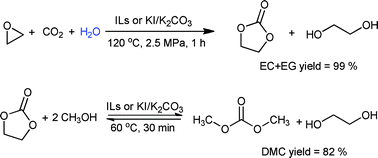
Synthesis of dimethyl carbonate from CO2 and ethylene oxide catalyzed by K2CO3-based binary salts in the presence of H2O - Green Chemistry (RSC Publishing)


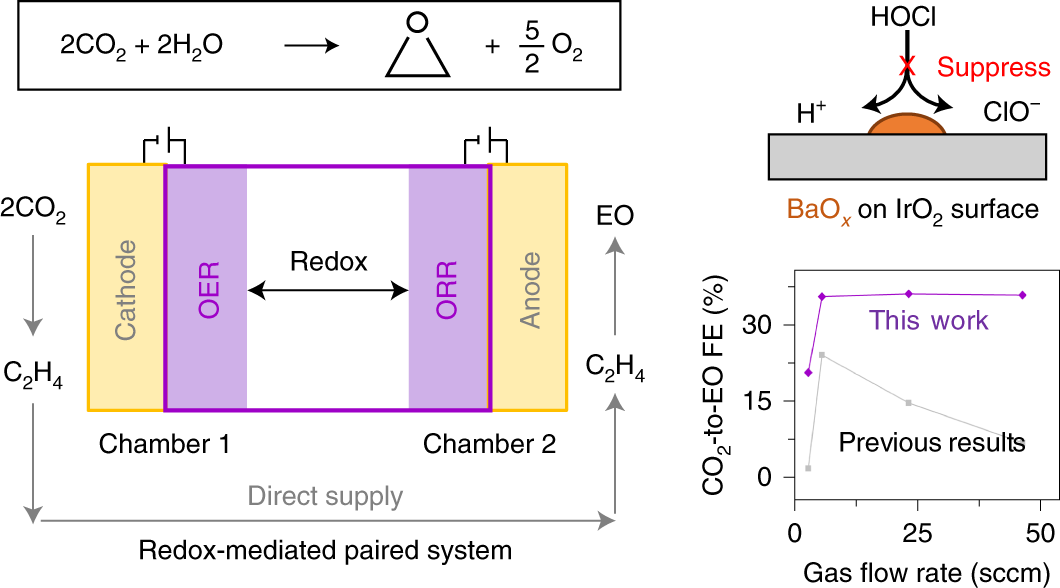




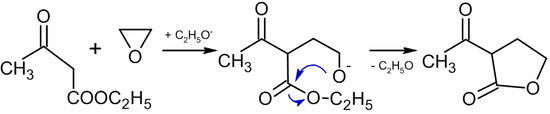
.jpg)
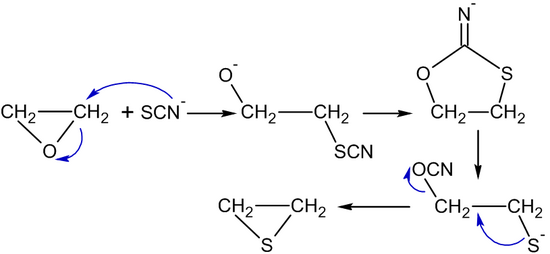
.jpg)




.jpg)
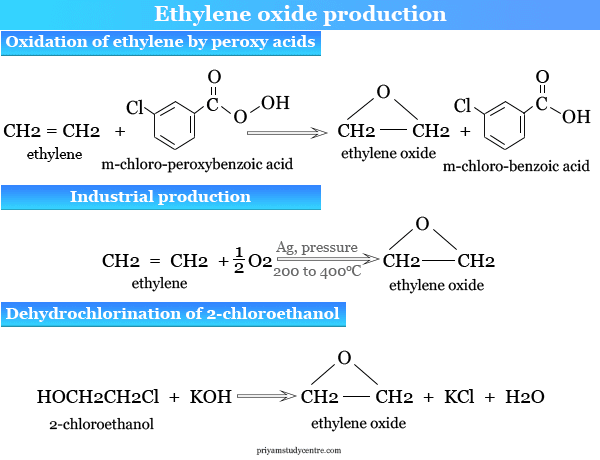
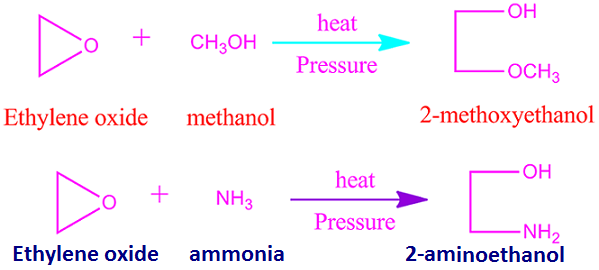
.jpg)