Ethanol, 25 l, plastic, CAS No. 64-17-5 | Acid Value Determination | Analysis of Food | Inorganic & Analytical Reagents | Chemicals | Carl Roth - International
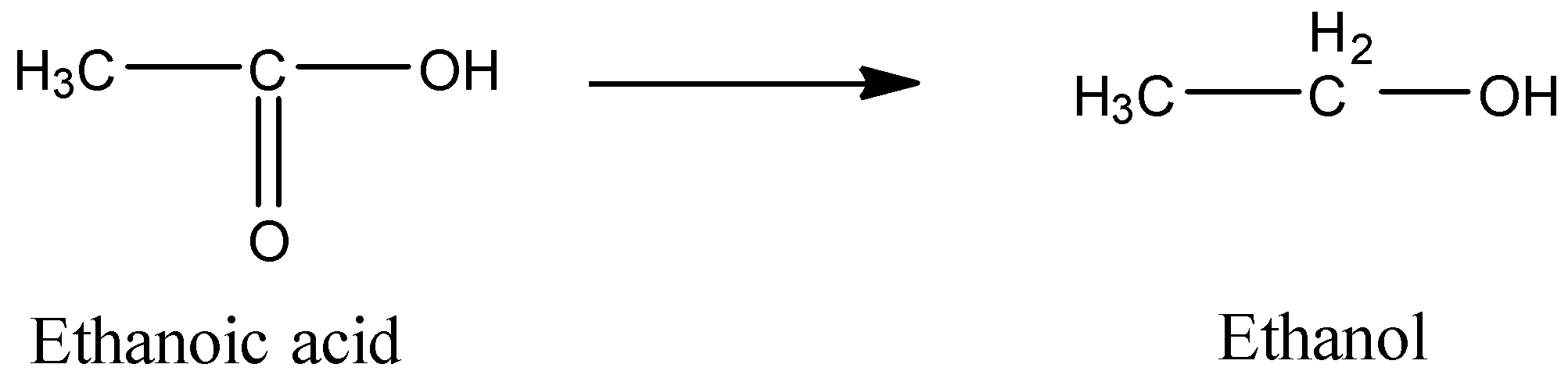
The reagent(s) used for converting ethanoic acid to ethanol is\/are:(A) $LiAl{H_4}$ (B) $B{H_3}$ in THF(C) $PC{l_3}$ (D) ${K_2}C{r_2}{O_7}\/{H^ + }$

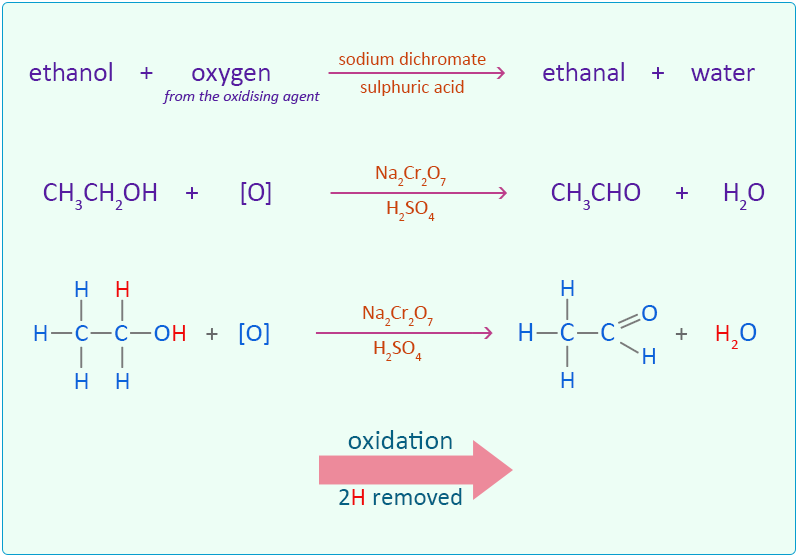
Question Video: Identifying the Class of Molecule of the Product of the Reaction of Ethanol with Concentrated Sulfuric Acid | Nagwa




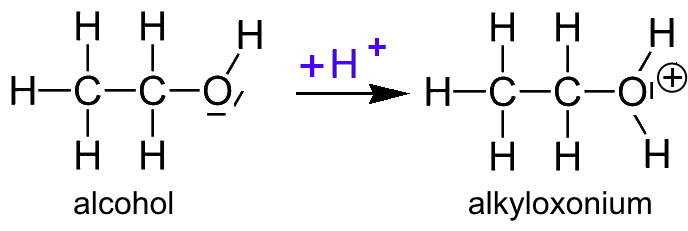



![Class 10] When ethanol reacts with ethanoic acid in presence of conc. Class 10] When ethanol reacts with ethanoic acid in presence of conc.](https://d1avenlh0i1xmr.cloudfront.net/b130c854-fc99-4f90-9538-20d0fd724d8b/reaction-of-ethanol-with-ethanoic-acid---teachoo.png)