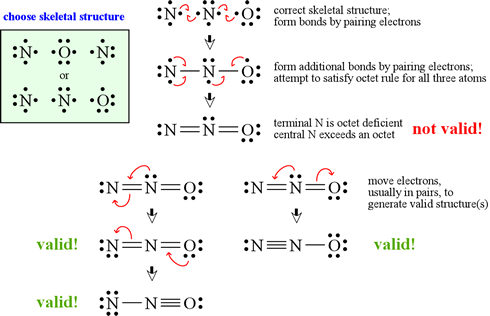
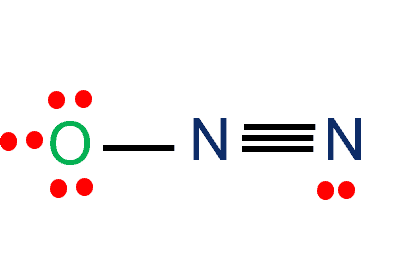
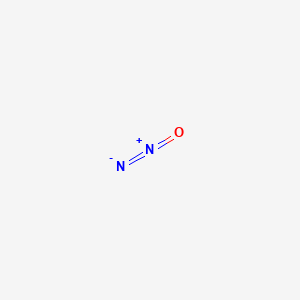
Reduction of Nitrous Oxide to Dinitrogen by a Mixed Valent Tricopper-Disulfido Cluster | Journal of the American Chemical Society

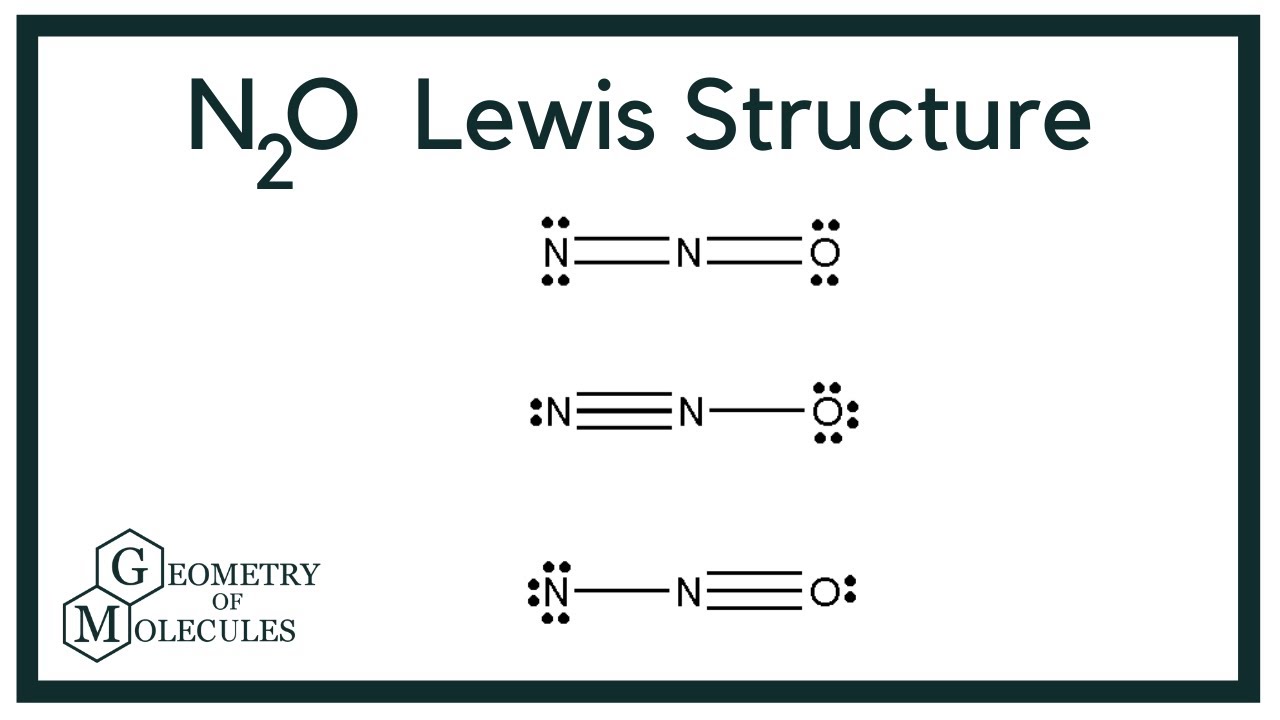
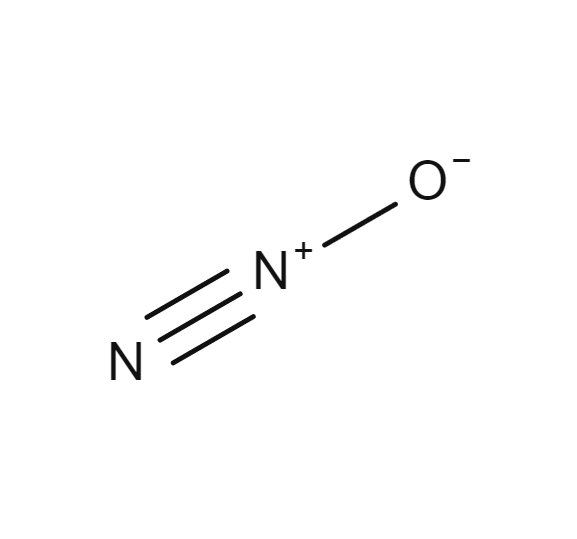
Dinitrogen monoxide (AKA nitrous oxide) reacts with oxygen gas according to the unbalanced equation N2O(g) + O2(g)→ NO2(g) Given the following legend and the reaction mixture shown below, answer the question that