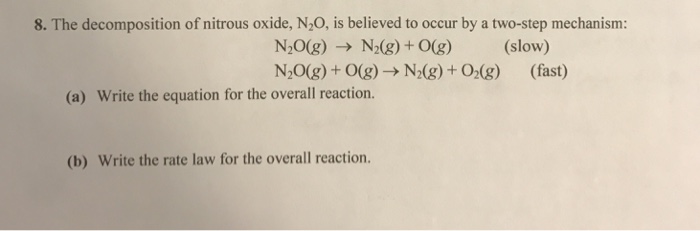SOLVED: The decomposition of nitrous oxide, N2O, is believed to occur by a two-step mechanism: N2O(g) → N2(g) + O(g) (slow) N2O(g) + O(g) → N2(g) + O2(g) (fast) (a) Write the
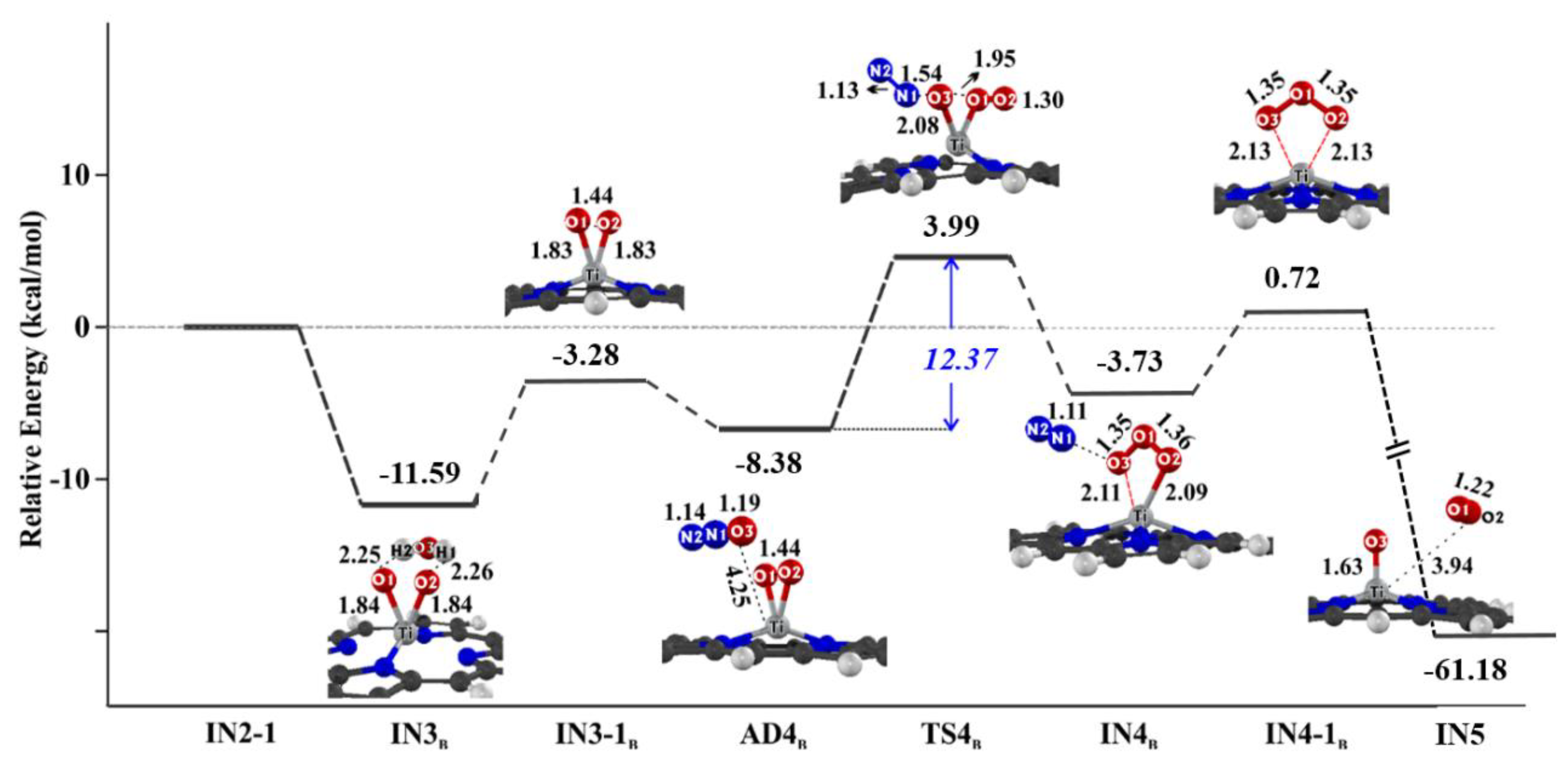
Catalysts | Free Full-Text | Effect of Water Molecule on Photo-Assisted Nitrous Oxide Decomposition over Oxotitanium Porphyrin: A Theoretical Study
![SOLVED: rate law for the decomposition of nitrous oxide (N2O) to Nz and 02 is Rate k[N2O] . Two 10. The experimental proposed: L ulld *o 0n mechanisms are 3 aNa 02 SOLVED: rate law for the decomposition of nitrous oxide (N2O) to Nz and 02 is Rate k[N2O] . Two 10. The experimental proposed: L ulld *o 0n mechanisms are 3 aNa 02](https://cdn.numerade.com/ask_images/210589257a0c4492a21a5f8ee5a0a594.jpg)
SOLVED: rate law for the decomposition of nitrous oxide (N2O) to Nz and 02 is Rate k[N2O] . Two 10. The experimental proposed: L ulld *o 0n mechanisms are 3 aNa 02
✓ Solved: Sketch a potential-energy diagram for the decomposition of nitrous oxide. N2O(g)→ N2(g)+O(g)...

On the mechanism of high-temperature N2O decomposition over Fe-FER in the presence of NO - ScienceDirect
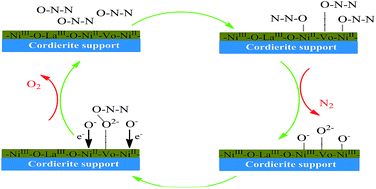
Supported Ni–La–Ox for catalytic decomposition of N2O I: component optimization and synergy - RSC Advances (RSC Publishing)

Molecules | Free Full-Text | N2O Decomposition over Fe-ZSM-5: A Systematic Study in the Generation of Active Sites
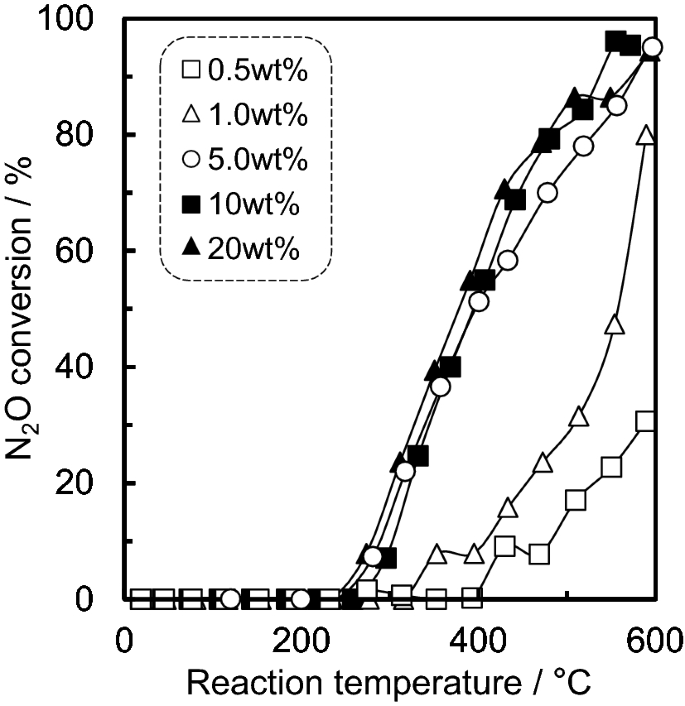
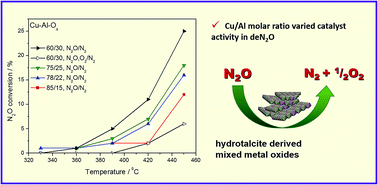
N2O decomposition properties of Ru catalysts supported on various oxide materials and SnO2 | Scientific Reports

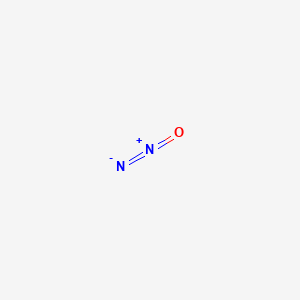
SOLVED:Nitrous oxide (N2 O), the "laughing gas" used as an anesthetic by dentists, is made by thermal decomposition of solid NH4 NO3 . Write a balanced equation for this reaction. What are
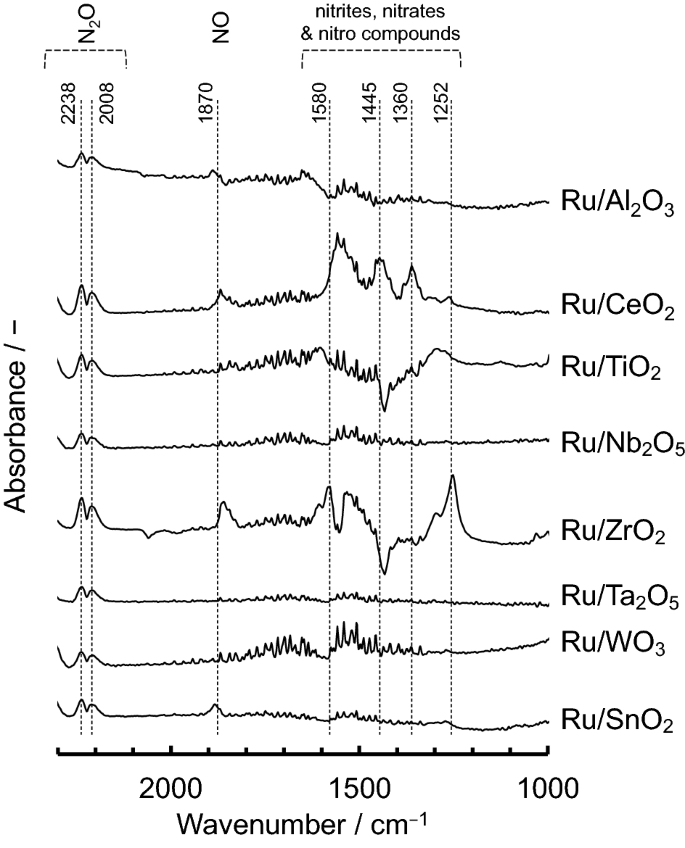
N2O decomposition properties of Ru catalysts supported on various oxide materials and SnO2 | Scientific Reports
![Data for the decomposition of dinitrogen oxide on a gold surface at 900 degrees Celsius are given below. Verify that the reaction is first order by preparing a graph of ln ~[N2O] Data for the decomposition of dinitrogen oxide on a gold surface at 900 degrees Celsius are given below. Verify that the reaction is first order by preparing a graph of ln ~[N2O]](https://homework.study.com/cimages/multimages/16/at16316449417038598771.jpg)