
Directional ionic transport across the oxide interface enables low-temperature epitaxy of rutile TiO2 | Nature Communications

Surface Segregation in Solid Oxide Cell Oxygen Electrodes: Phenomena, Mitigation Strategies and Electrochemical Properties | SpringerLink

Oxide Ion and Proton Conductivity in a Family of Highly Oxygen-Deficient Perovskite Derivatives | Journal of the American Chemical Society

Factors Governing Oxygen Vacancy Formation in Oxide Perovskites | Journal of the American Chemical Society
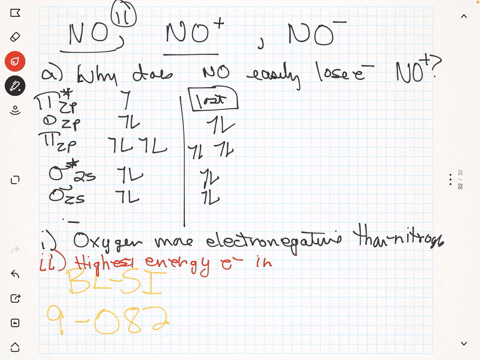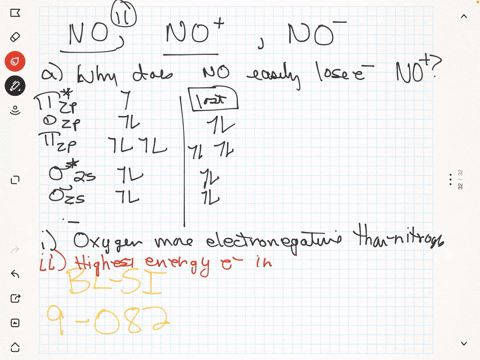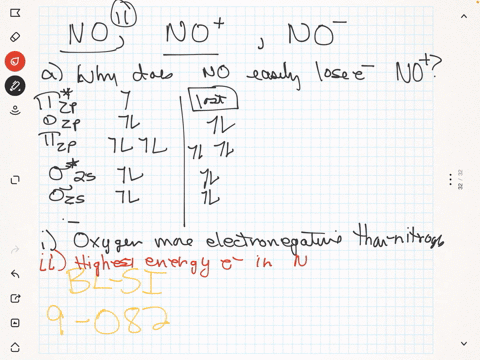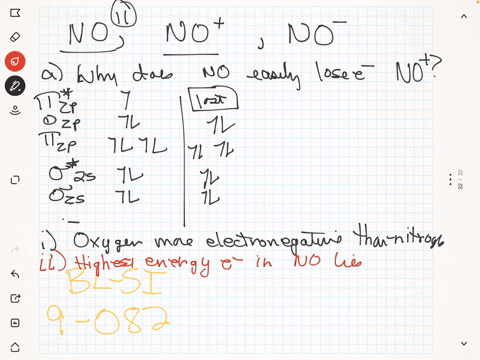
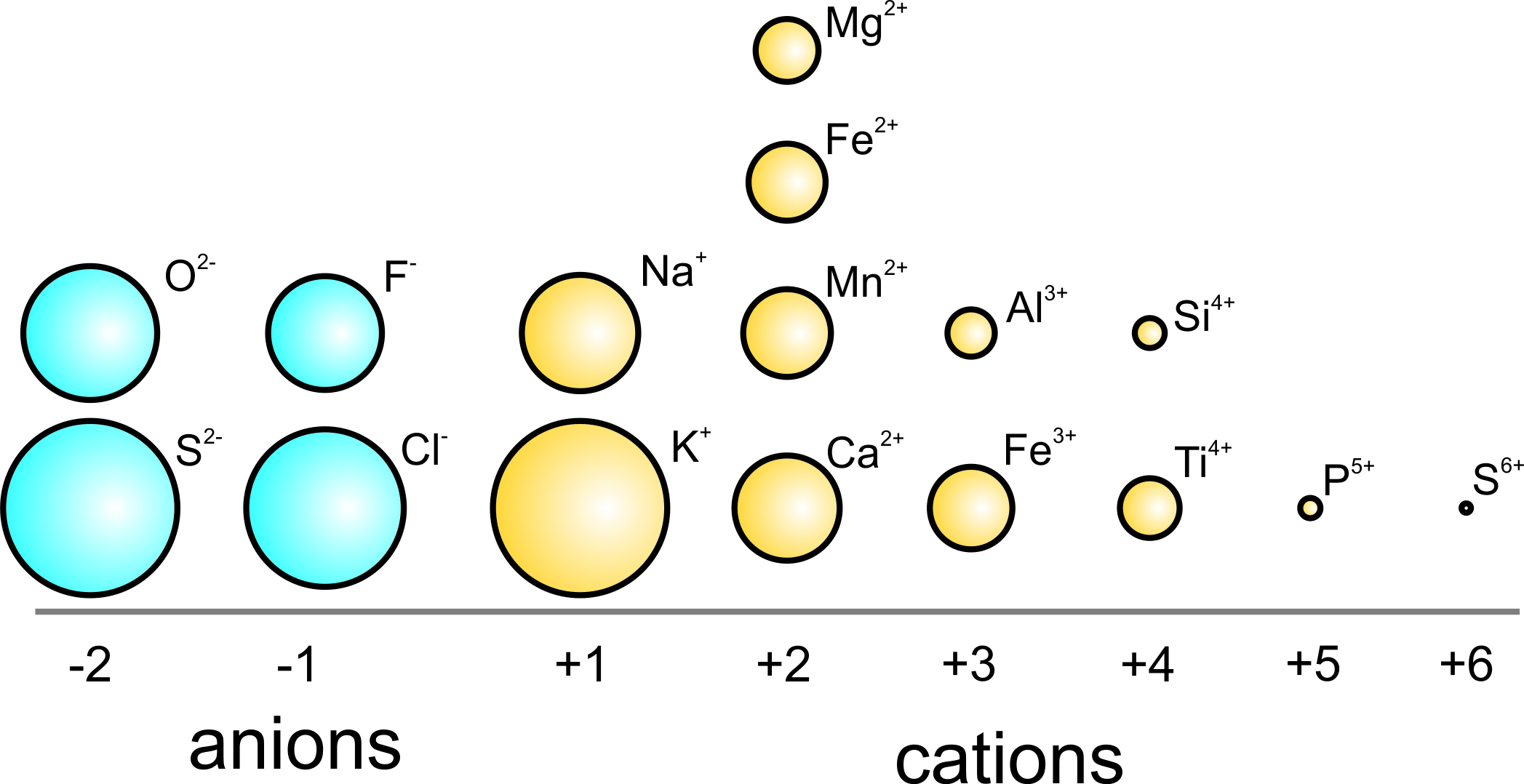
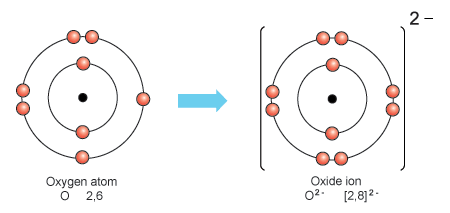
SOLVED: The ionic radius of the oxygen 2- anion is much different than that of the sulfur 2- anion. Choose the one answer that properly identifies their size difference and justifies its

Redox‐Inert Fe3+ Ions in Octahedral Sites of Co‐Fe Spinel Oxides with Enhanced Oxygen Catalytic Activity for Rechargeable Zinc–Air Batteries - Wang - 2019 - Angewandte Chemie International Edition - Wiley Online Library
Packing and the structural transformations in liquid and amorphous oxides from ambient to extreme conditions

SOLVED: The ionic radius of the oxygen 2- anion is much different than that of the sulfur 2- anion. Choose the one answer that properly identifies their size difference and justifies its

Catalysts | Free Full-Text | Factors Controlling the Redox Activity of Oxygen in Perovskites: From Theory to Application for Catalytic Reactions



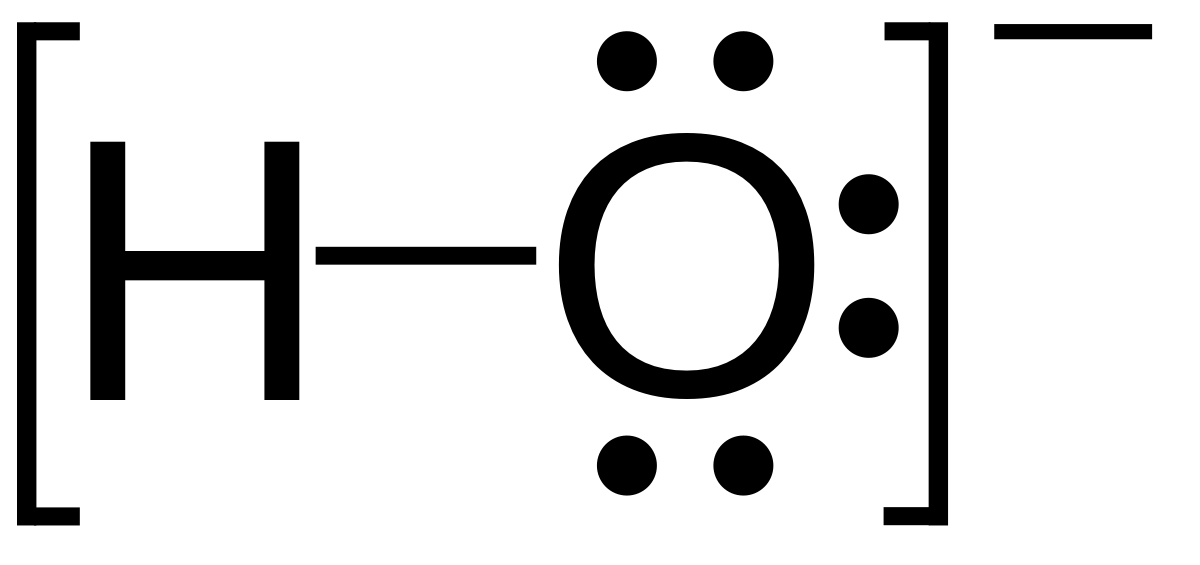


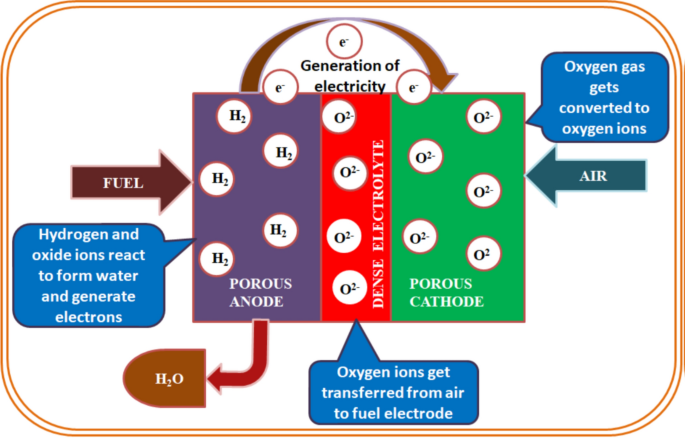
![Ion-oxygen parameters for various oxides[13] | Download Table Ion-oxygen parameters for various oxides[13] | Download Table](https://www.researchgate.net/publication/277646199/figure/tbl1/AS:613922981814311@1523382053524/on-oxygen-parameters-for-various-oxides13.png)


