
OneClass: 1. Copper metal (Cu) can be dissolved in nitric acid. The chemical equation for the reactio...

Concentrated Nitric Acid Reacting with Copper and Generating Nitrogen Dioxide Gas (decomposition of nitric acid producing gas Stock Photo - Alamy

what happens when copper reacts with concentrated nitric acid give the balance chemical equation for - Brainly.in

When copper is treated with a certain concentration of nitric acid, nitric oxide and nitrogen dioxide are liberated in equal volumes according to the equation: xCu + yHNO3⟶ Cu (NO3)2 + NO +

SOLVED: Copper reacts with nitric acid according to the following reaction: 3Cu + 8 HNO3 >3 Cu(NO3) 2 +2NO + 4H2O0 If 2.41g of Cu is added to a container with 2.00
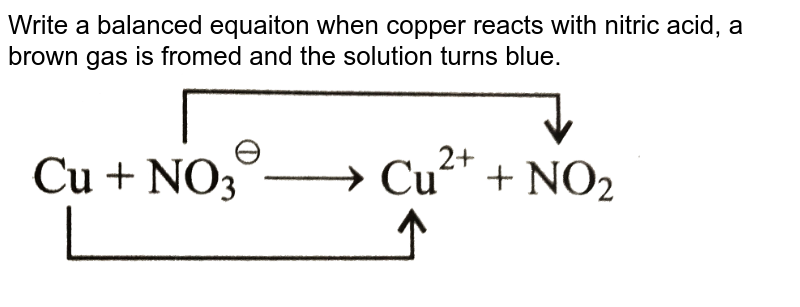
Copper reacts with nitric acid . A brown gas is formed and the solution turns blue. The equation may be written as : Cu+NO3^(-) rarr NO2+Cu^(2+) Balance the equation by oxidation number
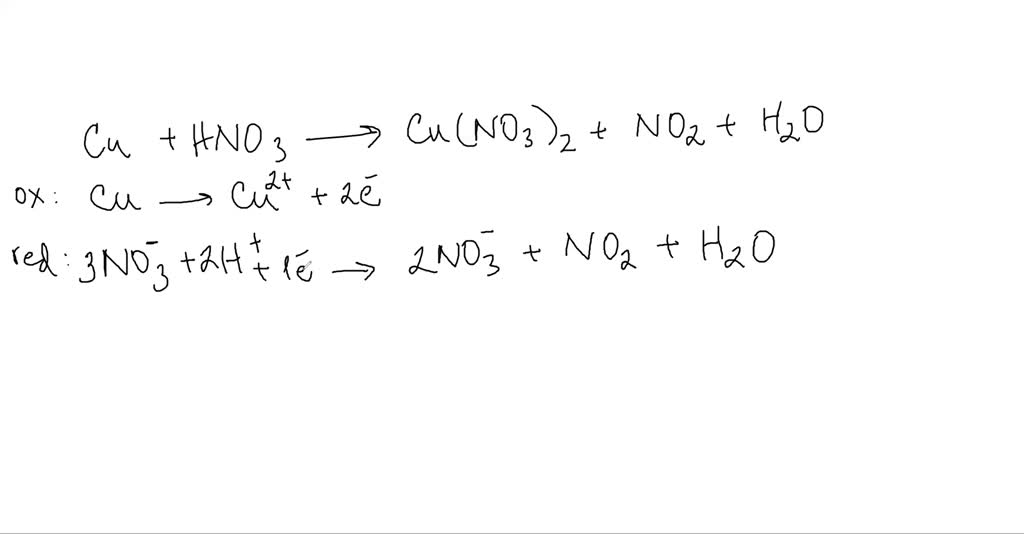


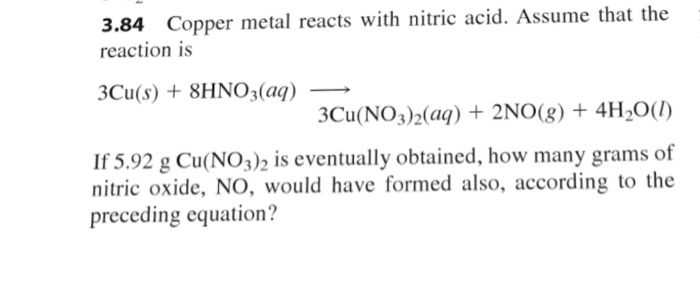


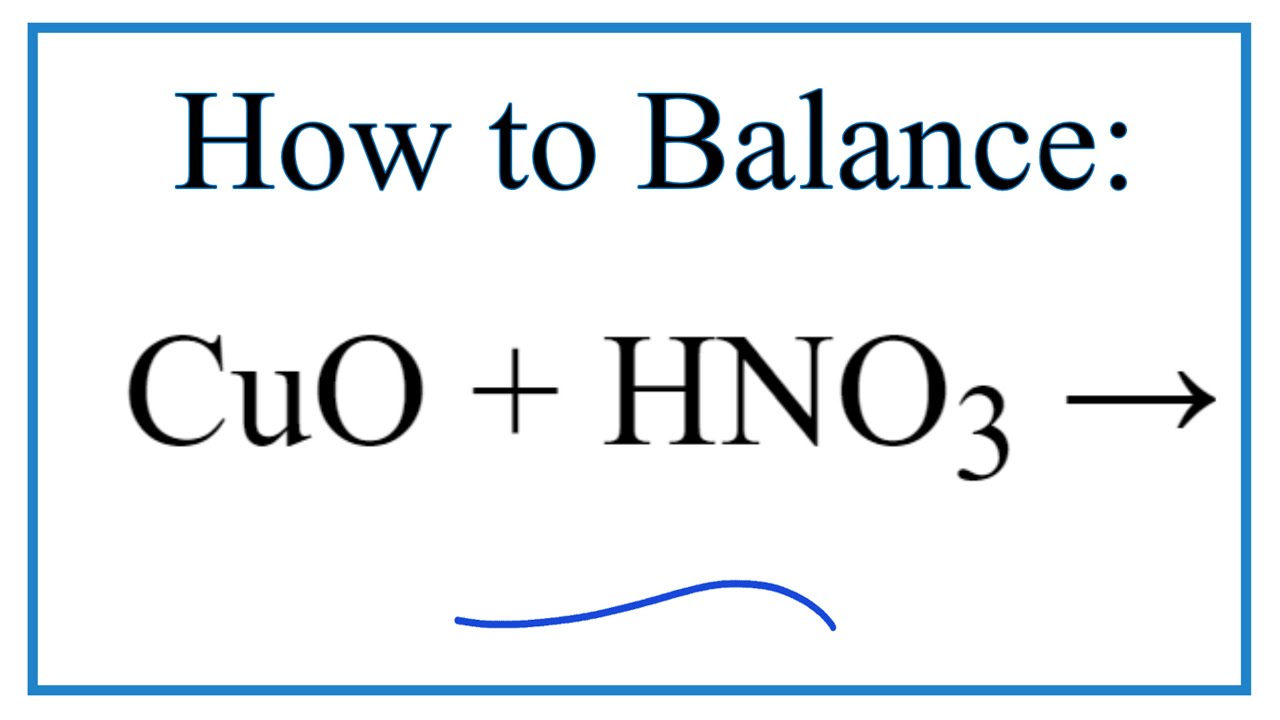

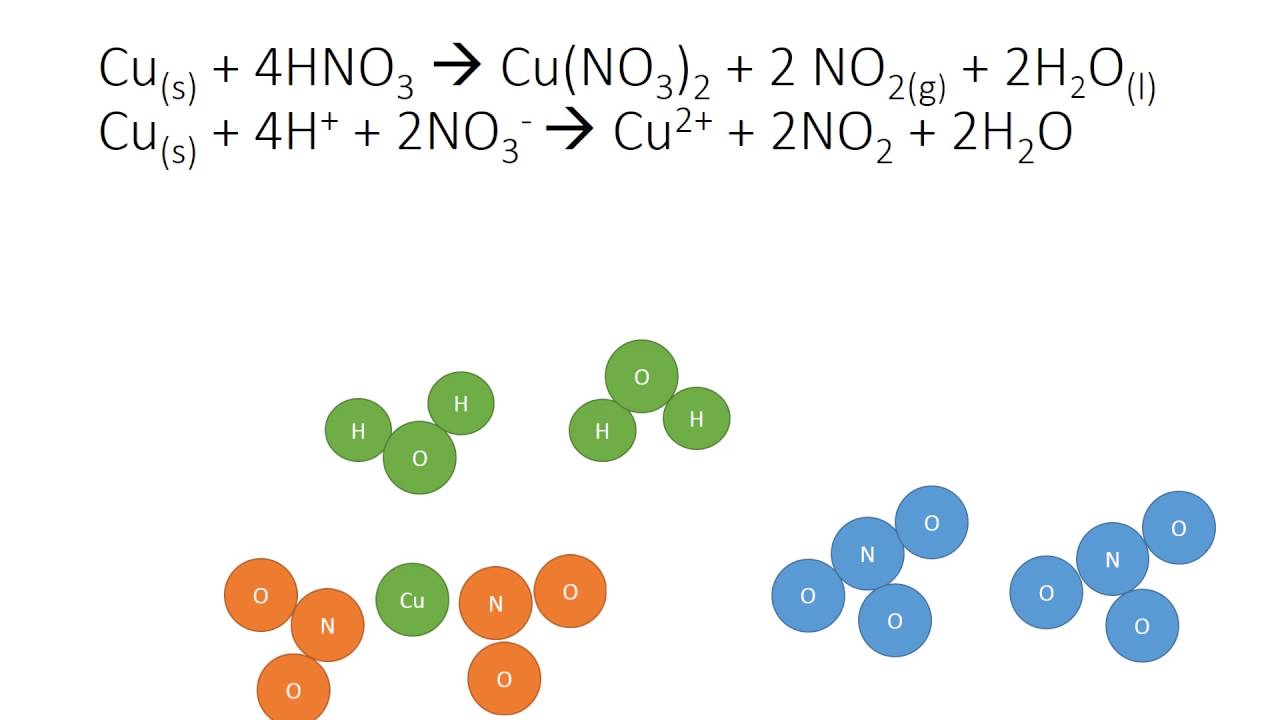








2%20+%20NO%20+%20H2O%20reaction.jpg?ezimgfmt=rs:323x202/rscb1/ngcb1/notWebP)


