Starter ručni poteznica trimeri Clover CG330 ,Farmer PRO 520,Iskra,Flora best, Villager BC900 | Permoto d.o.o.

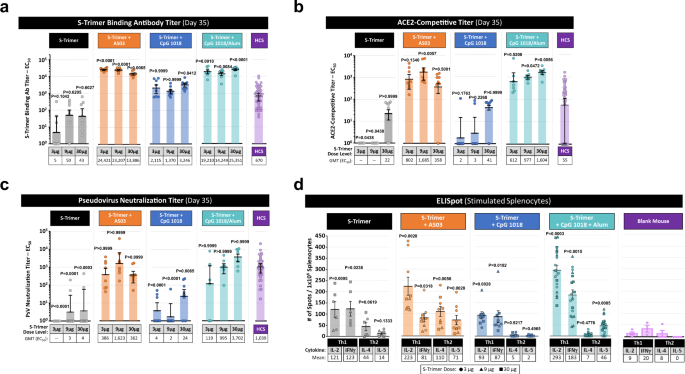
Clover Biopharmaceuticals Announces Publication of Phase 1 Clinical Trial Data for its Adjuvanted COVID-19 Vaccine Candidates in The Lancet

Safety and immunogenicity of a recombinant tandem-repeat dimeric RBD-based protein subunit vaccine (ZF2001) against COVID-19 in adults: two randomised, double-blind, placebo-controlled, phase 1 and 2 trials - The Lancet Infectious Diseases

Safety and immunogenicity of S-Trimer (SCB-2019), a protein subunit vaccine candidate for COVID-19 in healthy adults: a phase 1, randomised, double-blind, placebo-controlled trial - The Lancet

Using GSK and Dynavax tech, Clover kick-starts COVID-19 vax trial with data drop in August | FierceBiotech

Clover and GSK announce research collaboration to evaluate coronavirus (COVID-19) vaccine candidate with pandemic adjuvant system | GSK
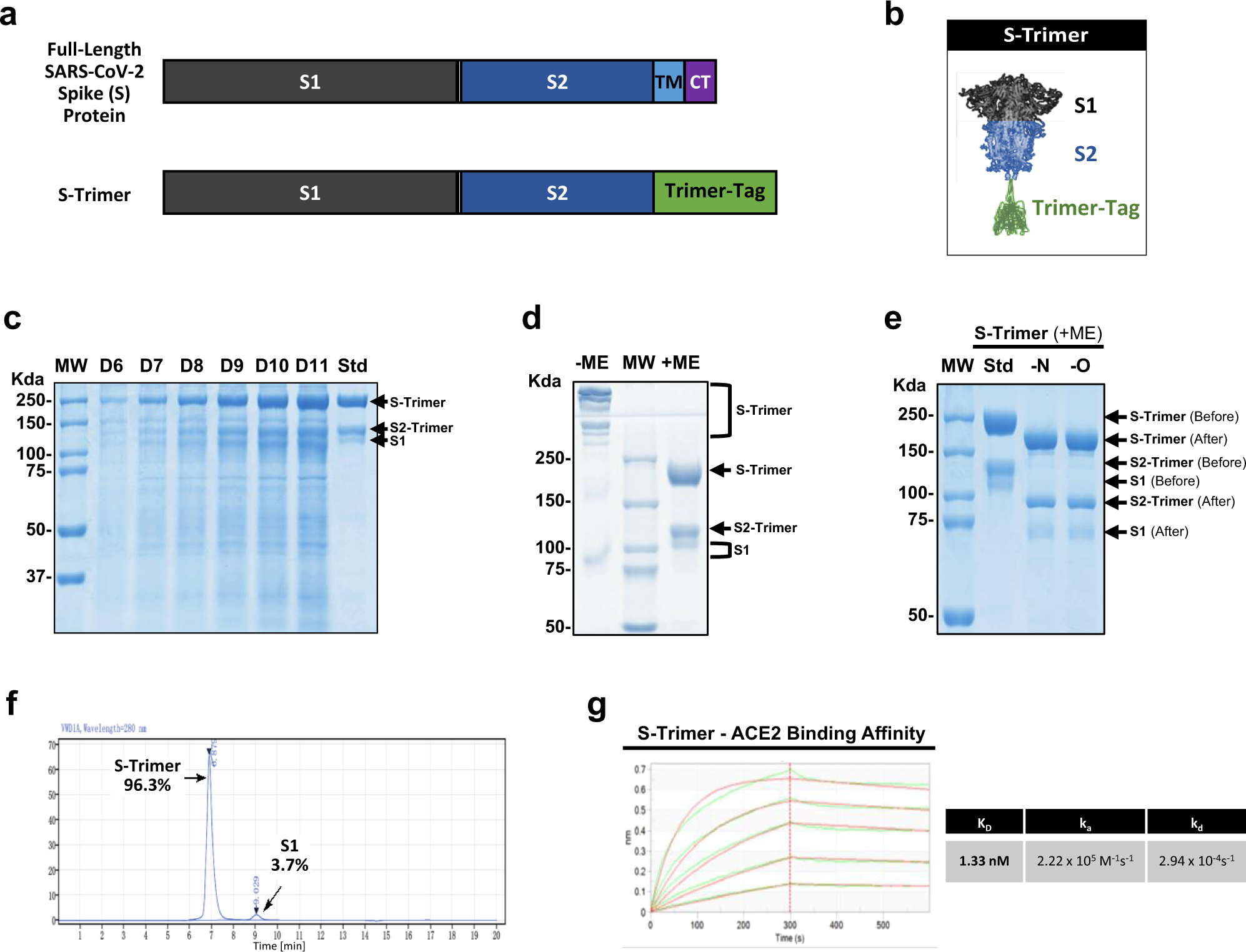
S-Trimer, a COVID-19 subunit vaccine candidate, induces protective immunity in nonhuman primates | Nature Communications
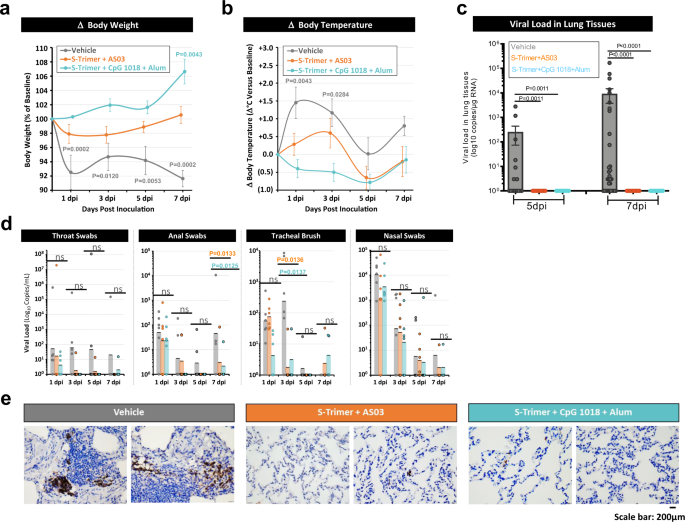
S-Trimer, a COVID-19 subunit vaccine candidate, induces protective immunity in nonhuman primates | Nature Communications
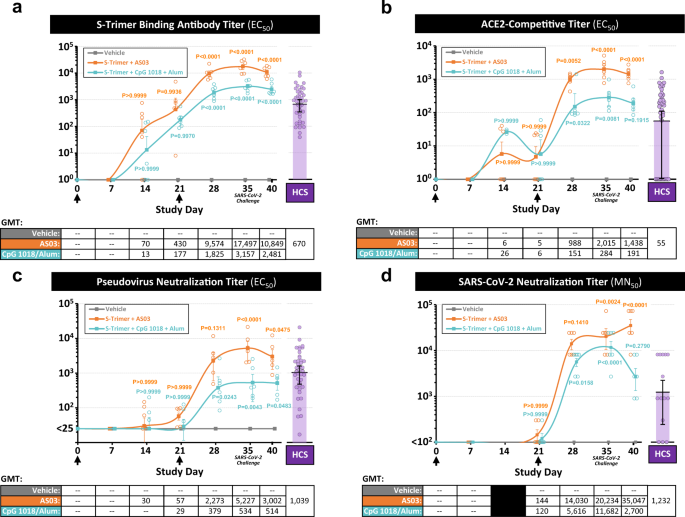
S-Trimer, a COVID-19 subunit vaccine candidate, induces protective immunity in nonhuman primates | Nature Communications

Clover Successfully Produced 2019-nCoV Subunit Vaccine Candidate and Detected Cross-Reacting Antibodies from Sera of Multiple Infected Patients | Business Wire