Write the net ionic reaction that occurs upon the addition of hno3 to a solution which contains methylamine - Brainly.com

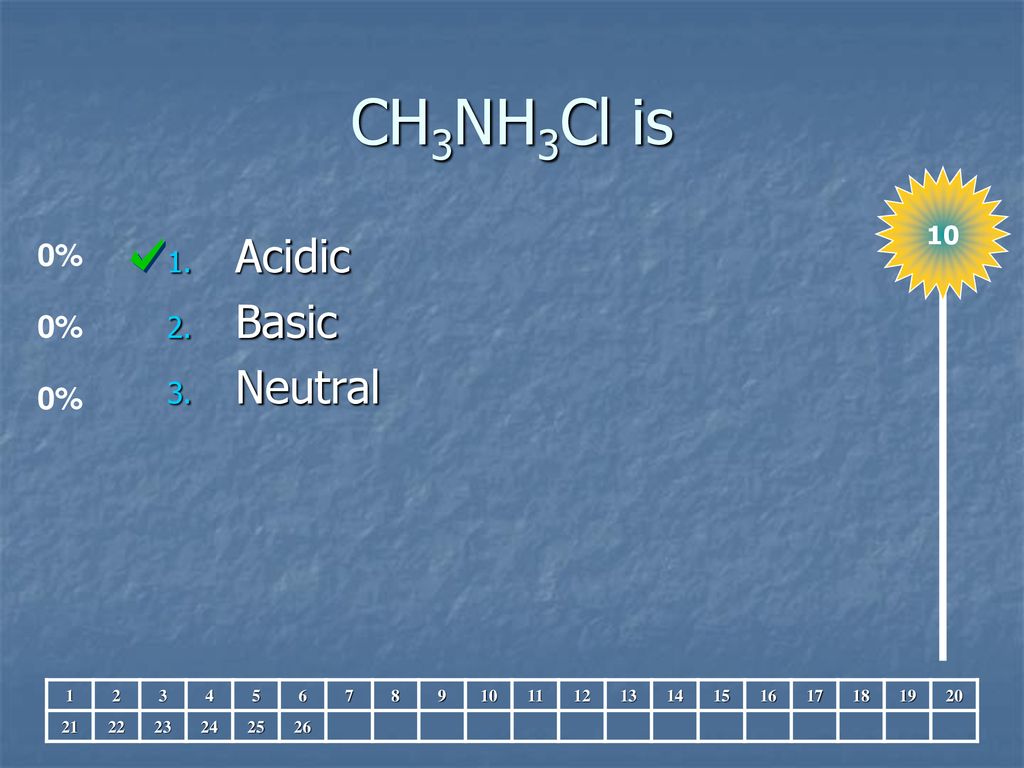
SOLVED: Which of the following compounds, when dissolved in water will produce an acidic solution? X. KNO3 Y. CH3NH3Cl Z. NaHSO4 X only X and Y Y and Z (Correct answer) Z

Which of the following mixtures would result in a buffered solution when 1.0 L of each of the two solutions are mixed? Why? : r/chemhelp

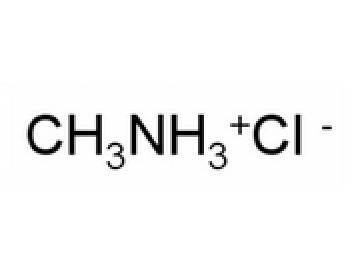
SOLVED: Methylamine, CH3NH2, and methylammonium chloride, CH3NH3Cl write the identity of the two species in each set (strong acid/strong base/weak acid/weak base/salt/other) and explain reason. Identify if anything in common betweenthe two

CNX Chemistry SSM Ch14 Mod06 - OpenStax College Chemistry 14: Buffers Chemistry 14: Acid-Base - Studocu

CH3NH3Cl-Assisted One-Step Solution Growth of CH3NH3PbI3: Structure, Charge-Carrier Dynamics, and Photovoltaic Properties of Perovskite Solar Cells | The Journal of Physical Chemistry C

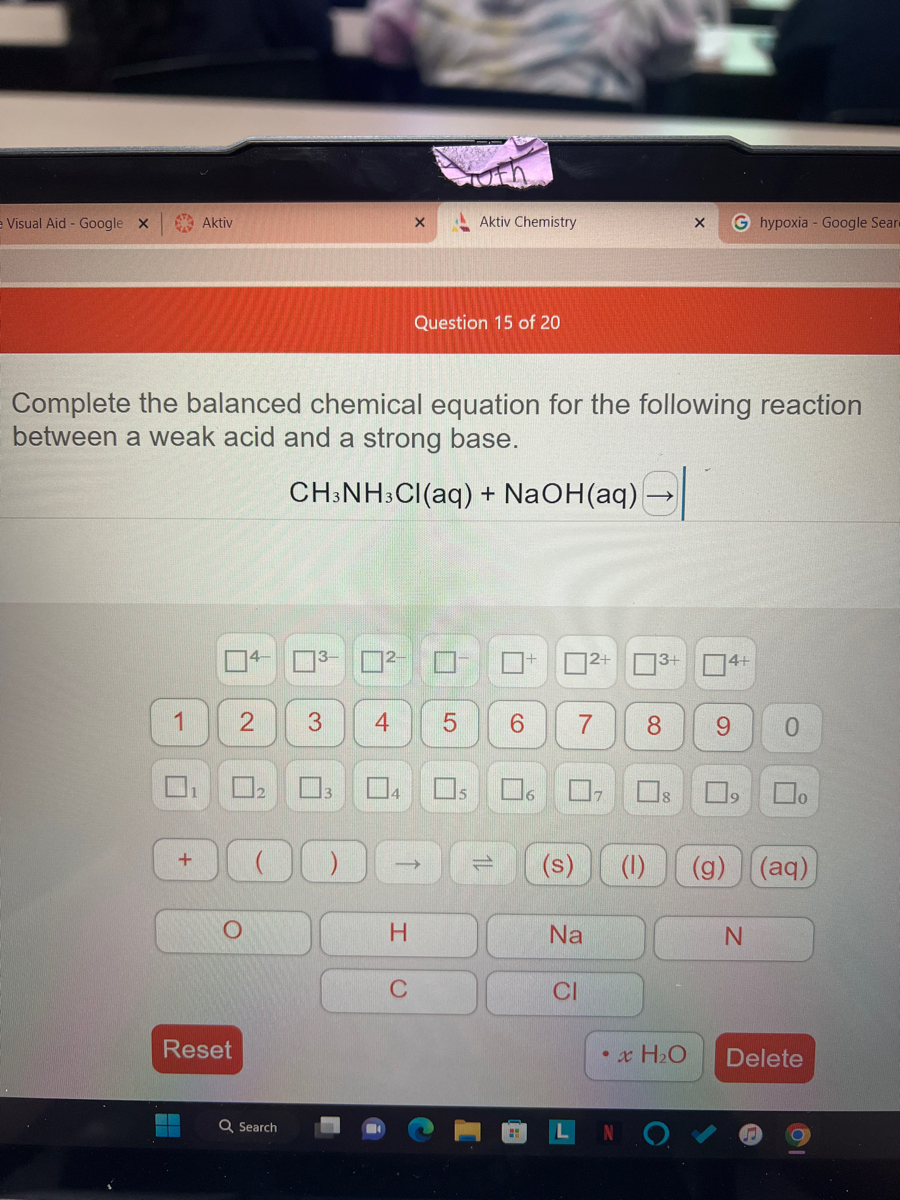
OneClass: please write an EQUILIBRIUM reaction that shows CH3NH3Cl asacidic- Write a chemical equatio...

OneClass: 1. Fe(NO3)3 2. Ba(NO3)2 3. NaOCl 4. NH4I 5. NH4NO2 6. CH3NH3Cl ***I can find the correct an...