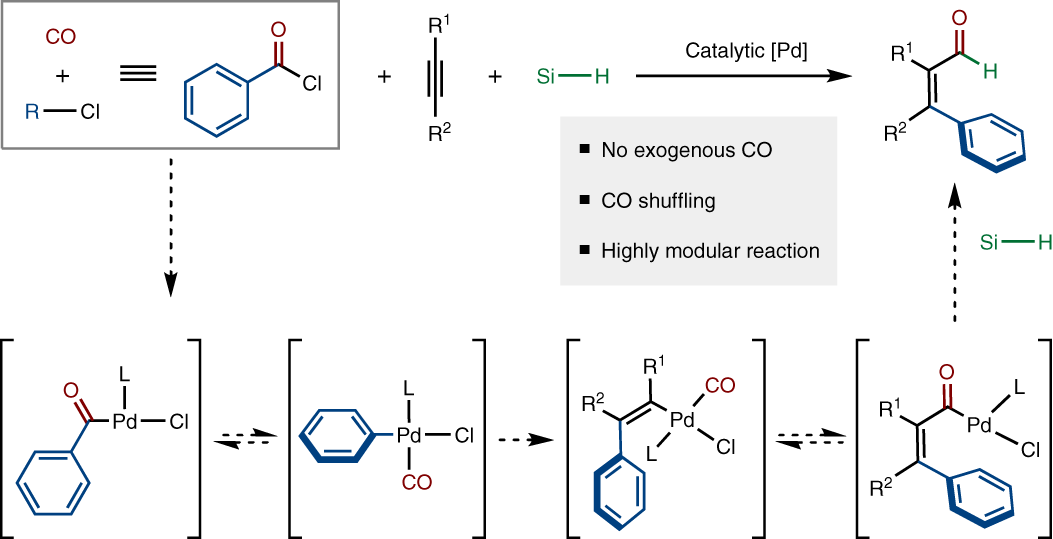
Synthesis and Insertion Reactions of the Cyclometalated Palladium−Alkyl Complexes Pd(CH2CMe2-o-C6H4)L2. Observation of a Pentacoordinated Intermediate in the Insertion of SO2 | Inorganic Chemistry

Crystal structure of the new palladium complexes tetrakis(1,3-dimethylimidazolium-2-ylidene)palladium(II) hexadecacarbonyltetrarhenium diethyl ether disolvate and octa-μ-carbonyl-dicarbonyltetrakis(triphenylphosphane)palladiumdirhenium (unknown solvate ...
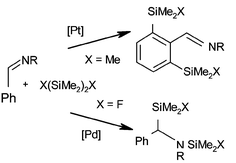
Palladium or platinum complex catalysed reactions of carbonyl and imine compounds with disilanes - Dalton Transactions (RSC Publishing)

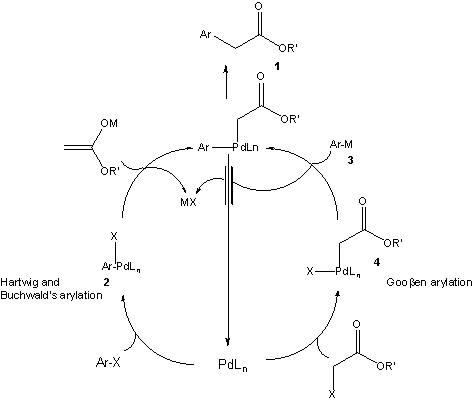
Palladium-catalyst development for the intermolecular carbonylative... | Download Scientific Diagram
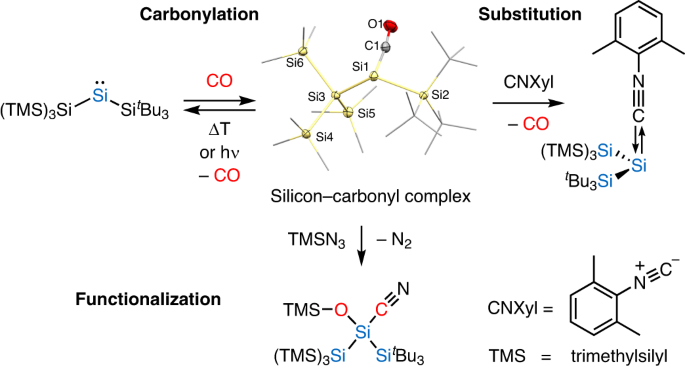
Silylated silicon–carbonyl complexes as mimics of ubiquitous transition-metal carbonyls | Nature Chemistry
Palladium(II) carboxylates and palladium(I) carbonyl carboxylate complexes as catalysts for olefin cyclopropanation with ethyl d

Reduction of α,β-unsaturated carbonyl compounds by palladium(II) and nickel(II) complexes having nitrogen-containing ligands - ScienceDirect
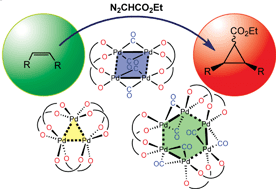
Palladium(II) carboxylates and palladium(I) carbonyl carboxylate complexes as catalysts for olefin cyclopropanation with ethyl diazoacetate - Dalton Transactions (RSC Publishing)

Direct Access to Palladium(II) Complexes Based on Anionic C,C,C-Phosphonium Ylide Core Pincer Ligand | Inorganic Chemistry

Gold(III)-CO and gold(III)-CO2 complexes and their role in the water-gas shift reaction | Science Advances

Bulky N‐Heterocyclic‐Carbene‐Coordinated Palladium Catalysts for 1,2‐Addition of Arylboron Compounds to Carbonyl Compounds - Okuda - 2020 - ChemCatChem - Wiley Online Library

Binuclear Palladium Complex Immobilized on Mesoporous SBA-16: Efficient Heterogeneous Catalyst for the Carbonylative Suzuki Coupling Reaction of Aryl Iodides and Arylboronic Acids Using Cr(CO)6 as Carbonyl Source | SpringerLink

Palladium-Catalyzed Cascade Carbonylative Synthesis of Perfluoroalkyl and Carbonyl-Containing 3,4-Dihydroquinolin-2(1H)-one Derivatives | Organic Letters

Diastereoselective Carbonyl Allylation with Simple Olefins Enabled by Palladium Complex-Catalyzed C-H Oxidative Borylation
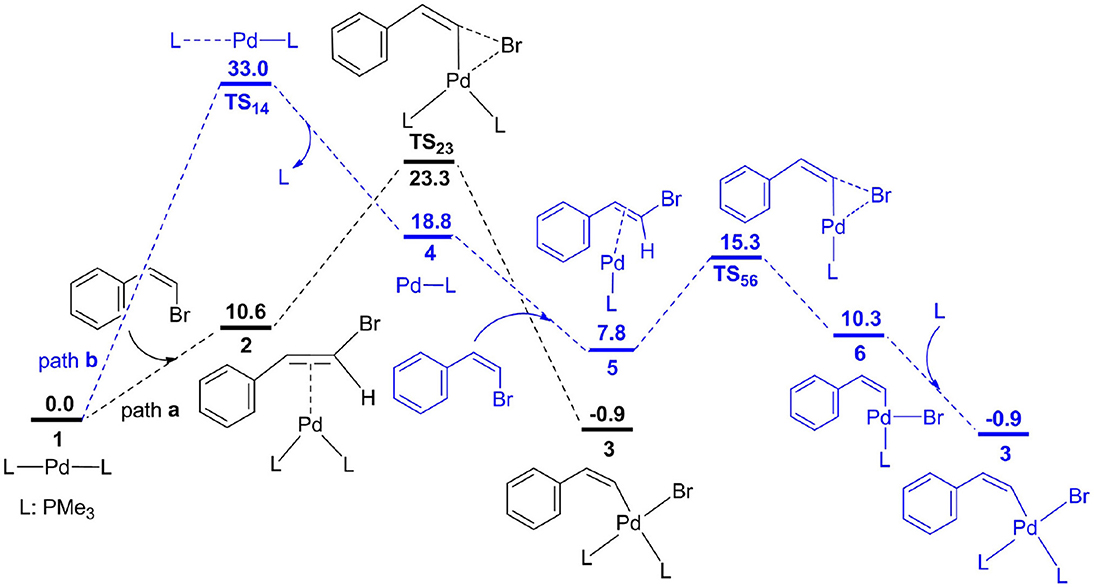
Frontiers | Key Mechanistic Features in Palladium-Catalyzed Methylcyclopropanation of Norbornenes With Vinyl Bromides: Insights From DFT Calculations

PDF) Reactivity of anionic metal carbonyl hydrides toward bis(diphenylphosphino)methane complexes of palladium and platinum | Pierre Braunstein - Academia.edu

Anchored Palladium Complex‐Generated Clusters on Zirconia: Efficiency in Reductive N‐Alkylation of Amines with Carbonyl Compounds under Hydrogen Atmosphere - Zhang - 2022 - Chemistry – An Asian Journal - Wiley Online Library