SOLVED: From the following formula calculate the quantity of each ingredient reuired to make 1500 g of the powder? Write answers in whole number Calcium Carbonate 5 parts magnesium oxide 1 parts

Limestone: Calcium Carbonate (CaCO3) - Uses, Preparation, Properties, Formula & Structure of Calcium Carbonate

Carbon dioxide is added to 112 g of calcium oxide. The product formed is 200 g of calcium carbonate. Calculate the mass of carbon dioxide used. Which law of chemical combination will

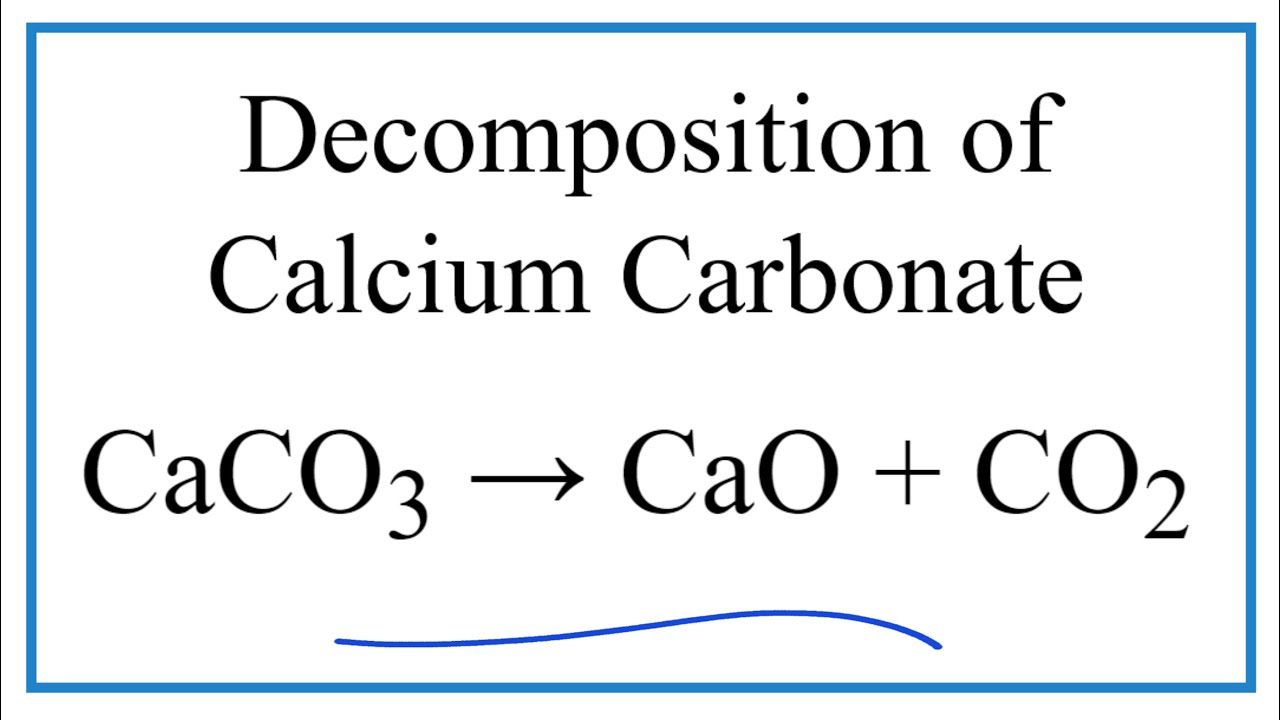
Metal carbonates are known to undergo thermal decomposition, producing the metal oxide and releasing carbon dioxide. The process is described by the following generic equation, in which M represents an unknown divalent

In Denmark the subsoil consists mainly of limestone. In contact with ground water containing carbon dioxide some of the calcium carbonate dissolves as calcium hydrogen carbonate. As a hydrogen carbonate causes problems

Sodium Oxide (Na2O) - Structure, Physical Properties, Chemical Properties and Uses with FAQs of Sodium Oxide