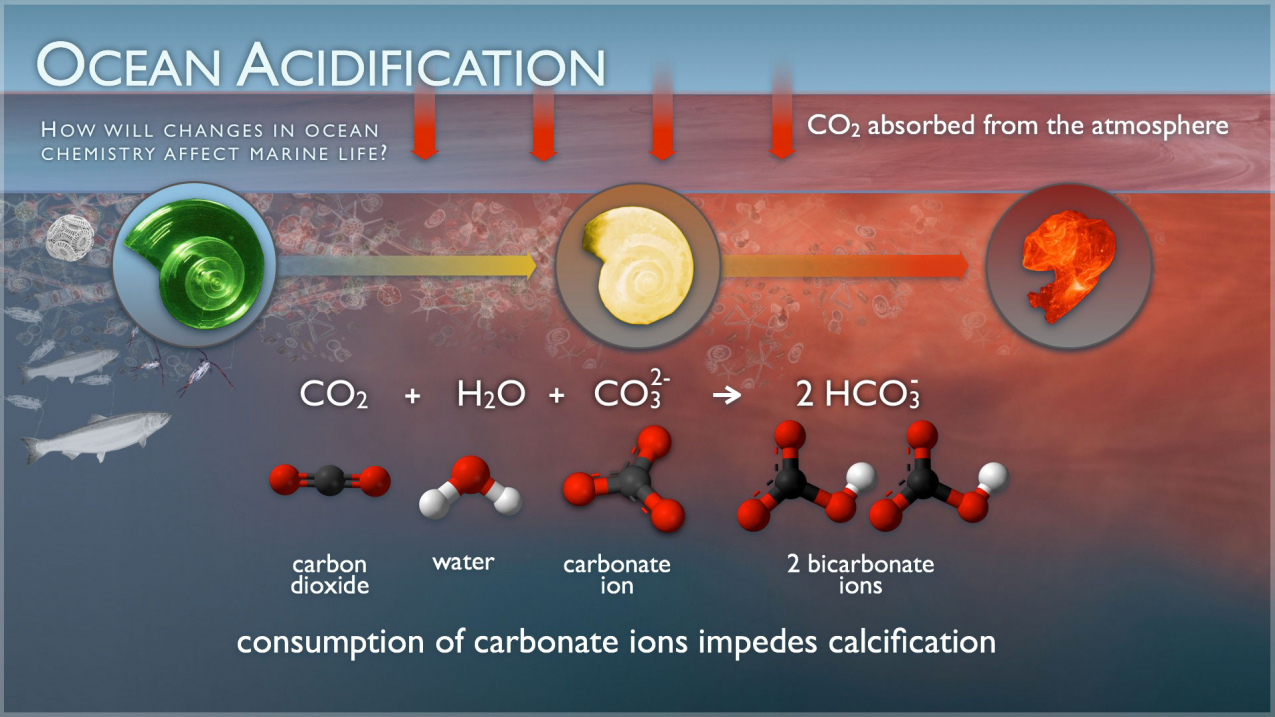
Carbonic Acid: An Important Intermediate in the Surface Chemistry of Calcium Carbonate | Journal of the American Chemical Society

Where does our drinking water come from? This means the water we drink has run through and across rocks. - ppt download
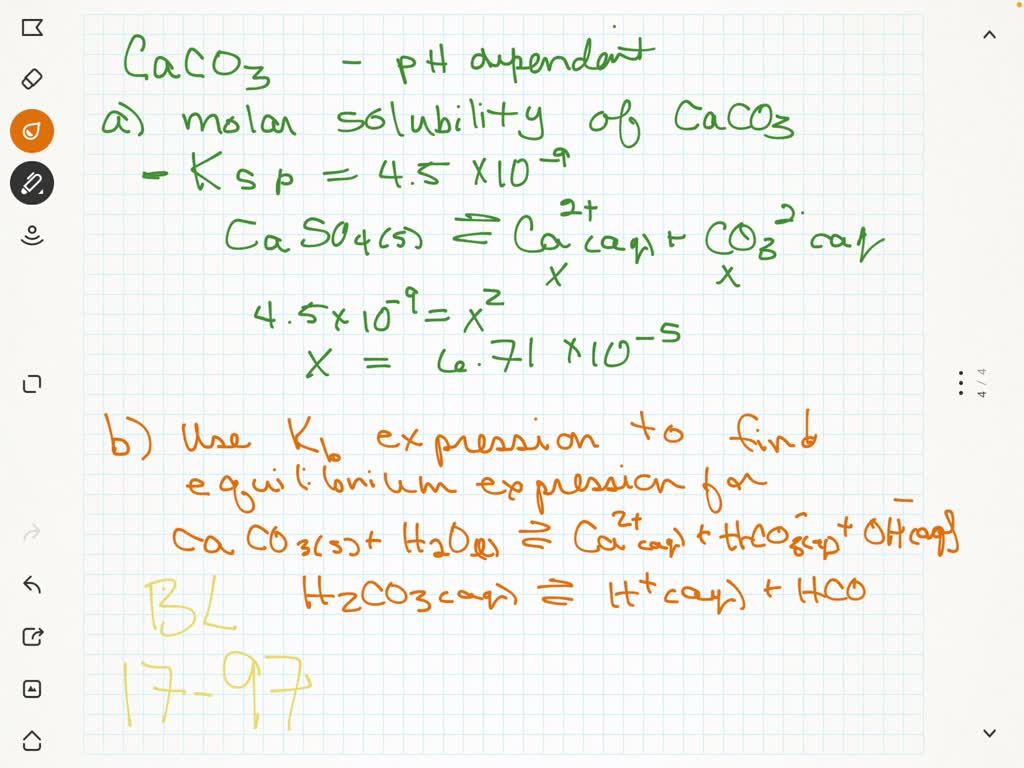
SOLVED: The KSP of CaCO3 is 3.369 × 10-9, the Ka of carbonic acid (H2CO3) is 4.400 × 10-7, the Ka of hydrogen carbonate ion (HCO3-) is 4.700 × 10-11, the Ka

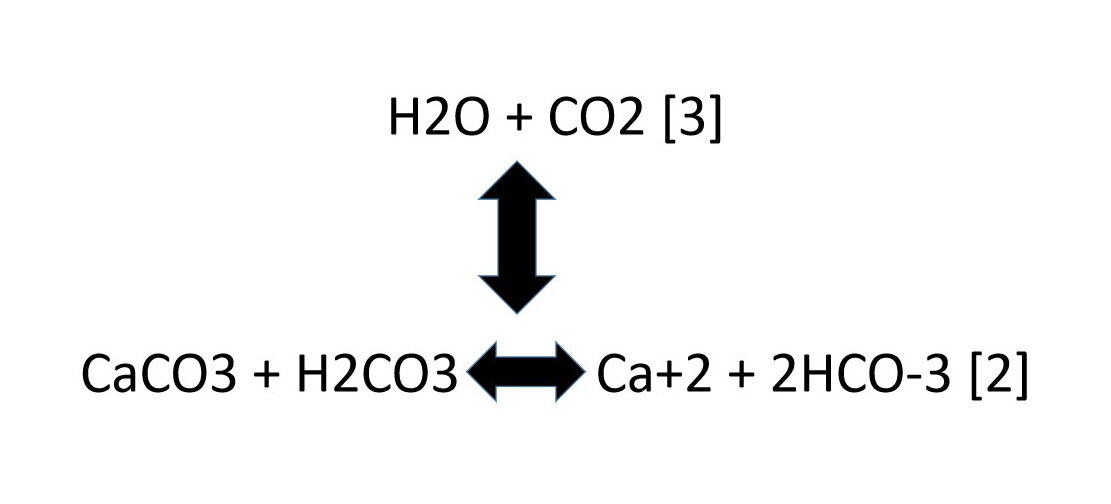
Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram