![4-(3-Methyl-benzoyl)-piperazin-1-yl]-acetic acid | CAS 705941-42-0 | SCBT - Santa Cruz Biotechnology 4-(3-Methyl-benzoyl)-piperazin-1-yl]-acetic acid | CAS 705941-42-0 | SCBT - Santa Cruz Biotechnology](https://media.scbt.com/product/4-3-methyl-benzoyl-piperazin-1-yl-acetic-acid-705941-42-0-_24_61_g_246188.jpg)
4-(3-Methyl-benzoyl)-piperazin-1-yl]-acetic acid | CAS 705941-42-0 | SCBT - Santa Cruz Biotechnology
![2-Oxo-4-[[[4-(2-aminoethyl)benzoyl]amino]acetyl]piperazine-1-acetic acid | Chemical Substance Information | J-GLOBAL 2-Oxo-4-[[[4-(2-aminoethyl)benzoyl]amino]acetyl]piperazine-1-acetic acid | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/009/215/J921.550F.png)
2-Oxo-4-[[[4-(2-aminoethyl)benzoyl]amino]acetyl]piperazine-1-acetic acid | Chemical Substance Information | J-GLOBAL
4-({4-[(Diaminomethylene)amino]benzoyl}oxy)phenyl]acetic acid methanesulfonate (1:1) | C17H19N3O7S | ChemSpider
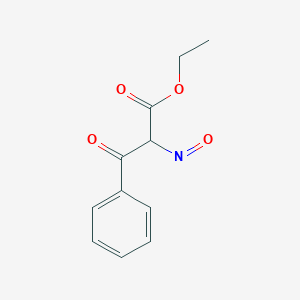
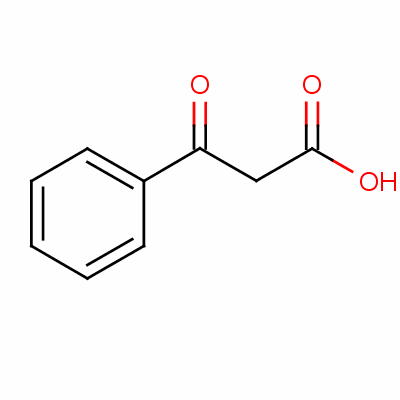
The product obtained when benzoyl acetic acid is heated with soda-lime is :(A) \n \n \n \n \n (B) \n \n \n \n \n (C) \n \n \n \n \n (D) \n \
The product obtained when benzoyl acetic acid is heated with soda-lime is :(A) \n \n \n \n \n (B) \n \n \n \n \n (C) \n \n \n \n \n (D) \n \
![2-[(2R,3R,5R)-3-Benzoyl-5-(4-bromophenyl)tetrahydrothiophene-2-yl]acetic acid ethyl ester | Chemical Substance Information | J-GLOBAL 2-[(2R,3R,5R)-3-Benzoyl-5-(4-bromophenyl)tetrahydrothiophene-2-yl]acetic acid ethyl ester | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/034/063/J3.406.360E.png)
2-[(2R,3R,5R)-3-Benzoyl-5-(4-bromophenyl)tetrahydrothiophene-2-yl]acetic acid ethyl ester | Chemical Substance Information | J-GLOBAL
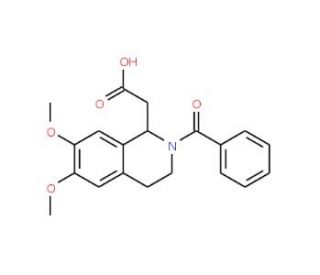
2-benzoyl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinolin-1-yl)acetic acid | SCBT - Santa Cruz Biotechnology
![2-[1-[4-[[4-(Trifluoromethyl)benzoyl]amino]benzoyl]isoquinolin-4-yl]acetic acid | C26H17F3N2O4 - PubChem 2-[1-[4-[[4-(Trifluoromethyl)benzoyl]amino]benzoyl]isoquinolin-4-yl]acetic acid | C26H17F3N2O4 - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=70691974&t=l)
2-[1-[4-[[4-(Trifluoromethyl)benzoyl]amino]benzoyl]isoquinolin-4-yl]acetic acid | C26H17F3N2O4 - PubChem
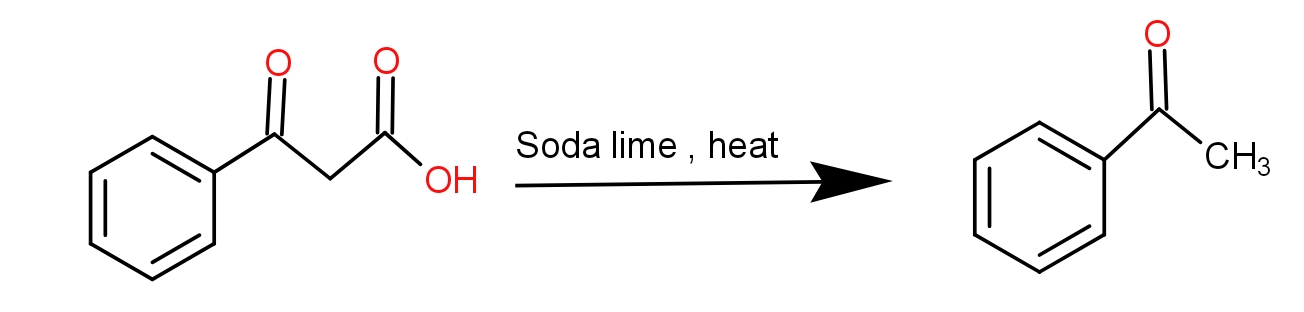
The product obtained when benzoyl acetic acid is heated with soda-lime is :(A) \n \n \n \n \n (B) \n \n \n \n \n (C) \n \n \n \n \n (D) \n \
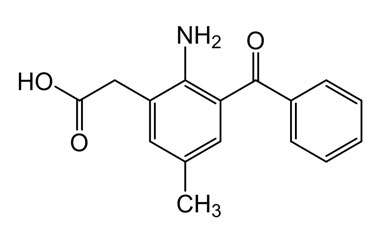
CAS No : 91714-75-9 | Product Name : 2-(2-Amino-3-benzoyl-5-methylphenyl)acetic Acid | Pharmaffiliates

Propose a mechanism for the reaction of benzoyl chloride with acetic acid, and show the structure of the resulting anhydride. | Homework.Study.com

![2-Cyano-ethyl)-(4-nitro-benzoyl)-amino]-acetic acid ethyl ester 2-Cyano-ethyl)-(4-nitro-benzoyl)-amino]-acetic acid ethyl ester](https://www.dougdiscovery.com/media/catalog/product/cache/743e8da6510972d27e930483041623da/I/P/IPFQBAJJAABEGL-UHFFFAOYSA-N_480236.png)



![1-(4-Chloro-benzoyl)-cyclohexyl]-acetic acid | C15H17ClO3 - PubChem 1-(4-Chloro-benzoyl)-cyclohexyl]-acetic acid | C15H17ClO3 - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=18412333&t=l)