
DIY Thermite Required Materials Iron Oxide aka rust Powdered Aluminum (these are both available from paint stores) Magnesium strip or sparkler Protective mask and gloves Scale Bucket heavy duty plastic bag Container
![PDF] Mechanism for thermite reactions of aluminum/iron-oxide nanocomposites based on residue analysis | Semantic Scholar PDF] Mechanism for thermite reactions of aluminum/iron-oxide nanocomposites based on residue analysis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/035f93a77120a19b7cb355ec122296f5319649ce/3-Figure1-1.png)
PDF] Mechanism for thermite reactions of aluminum/iron-oxide nanocomposites based on residue analysis | Semantic Scholar
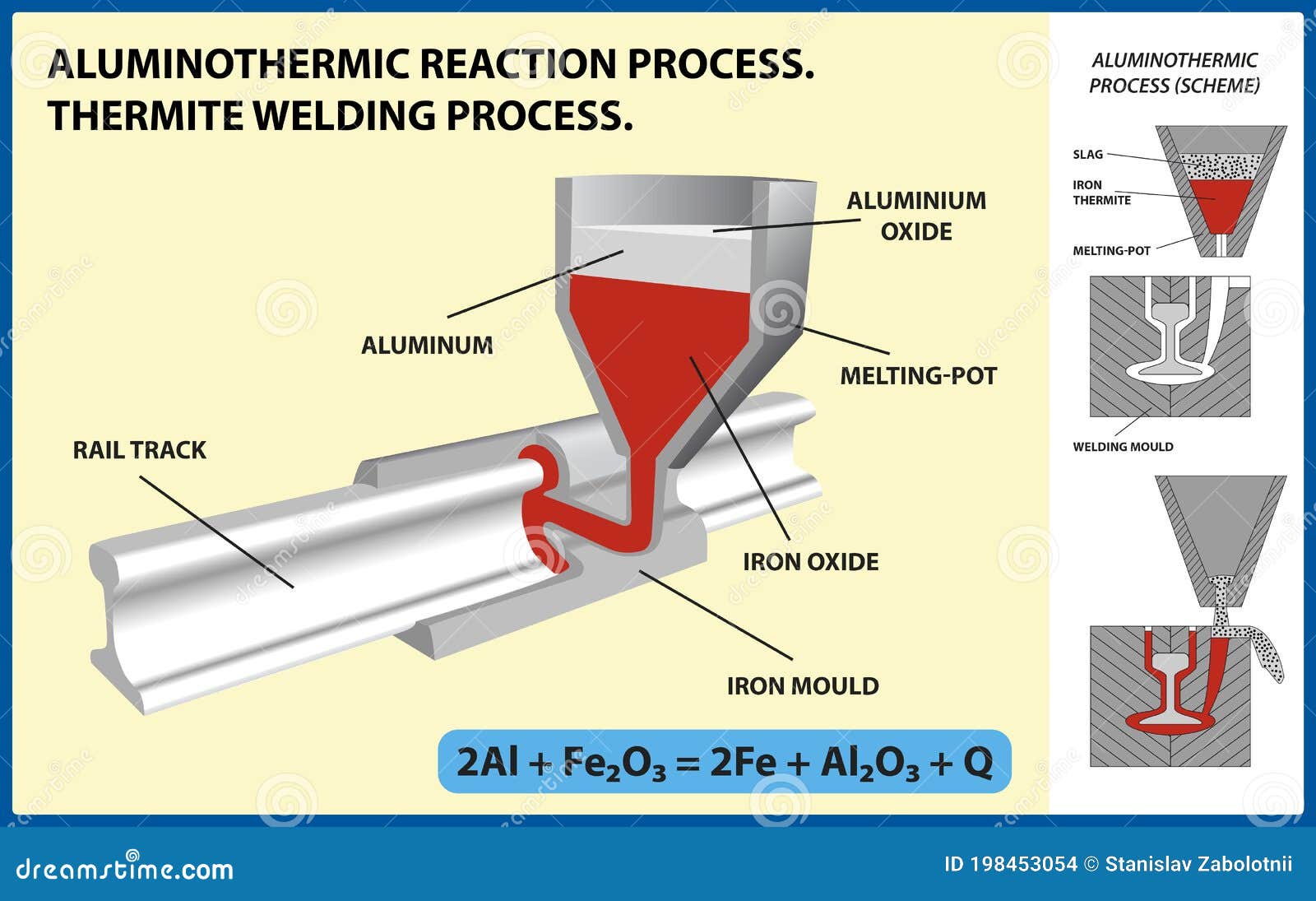
Aluminothermic Reaction Process. Thermite Welding Process. Vector Illustration Stock Vector - Illustration of evolved, mixture: 198453054

Stoichiometry AP Chemistry In the thermite reaction, a mixture of powdered aluminum and powdered iron(III) oxide react to yield iron and aluminum oxide. - ppt download

SOLVED:In the thermite reaction, iron(III) oxide is reduced by aluminum to give molten iron. Fe2 O3(s)+2 Al(s) →2 Fe(ℓ)+Al2 O3(s) If you begin with 10.0 g of Fe2 O3 and 20.0 g

SOLVED: The thermite reaction has solid aluminum powder (28.871 J(mol-K ) reacting with iron(Ill) oxide (87.793 J(mol-K to make aluminum oxide (49.56 J(mol-K ) and iron (25.305 J(mol:K) The reaction is So
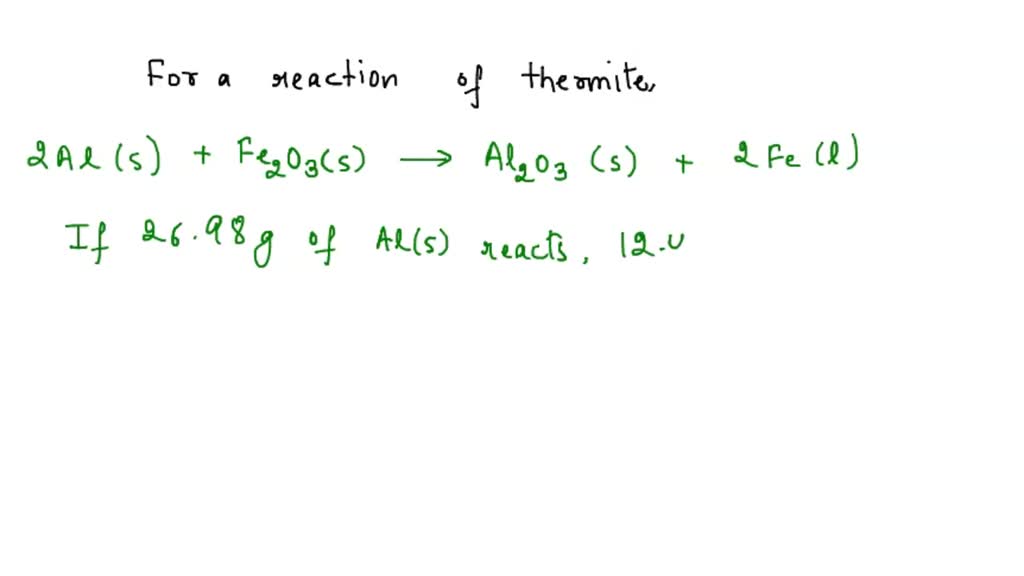










.jpg)






:max_bytes(150000):strip_icc()/GettyImages-578813868-5a787515a9d4f90036eff588.jpg)
