Hydrogen generation by electrolysis under subcritical water condition and the effect of aluminium anode - ScienceDirect
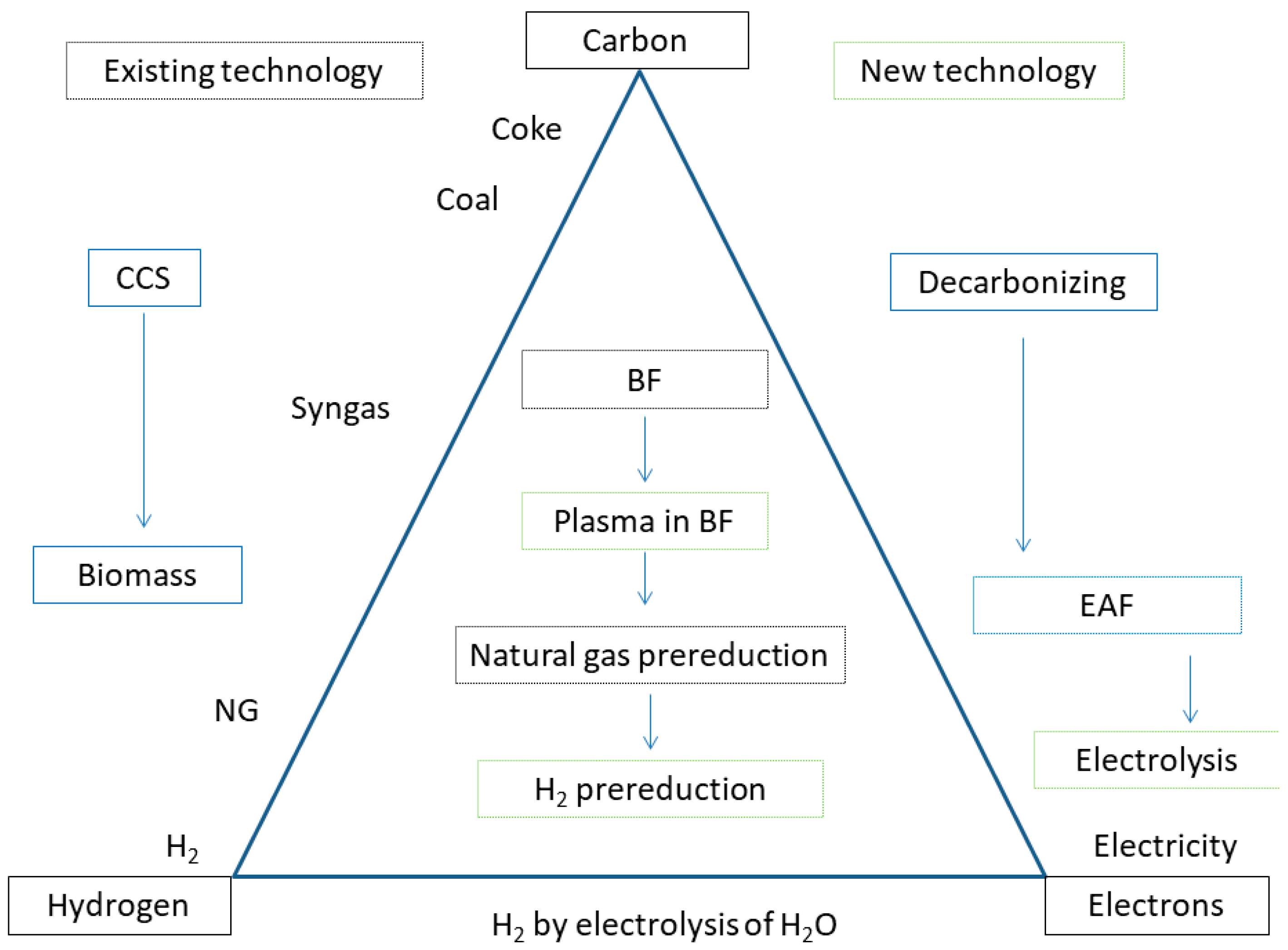
Metals | Free Full-Text | Water Electrolysis for the Production of Hydrogen to Be Employed in the Ironmaking and Steelmaking Industry

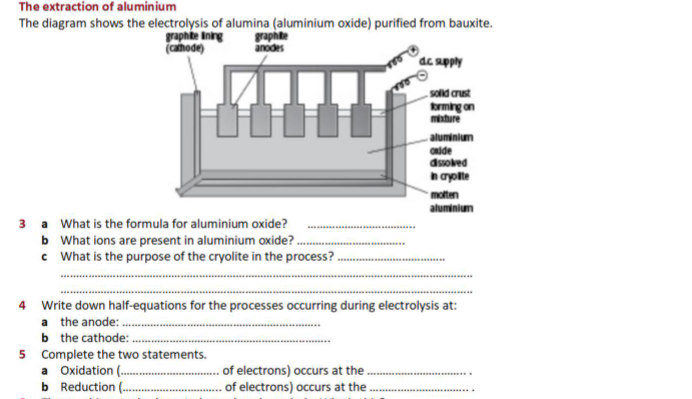
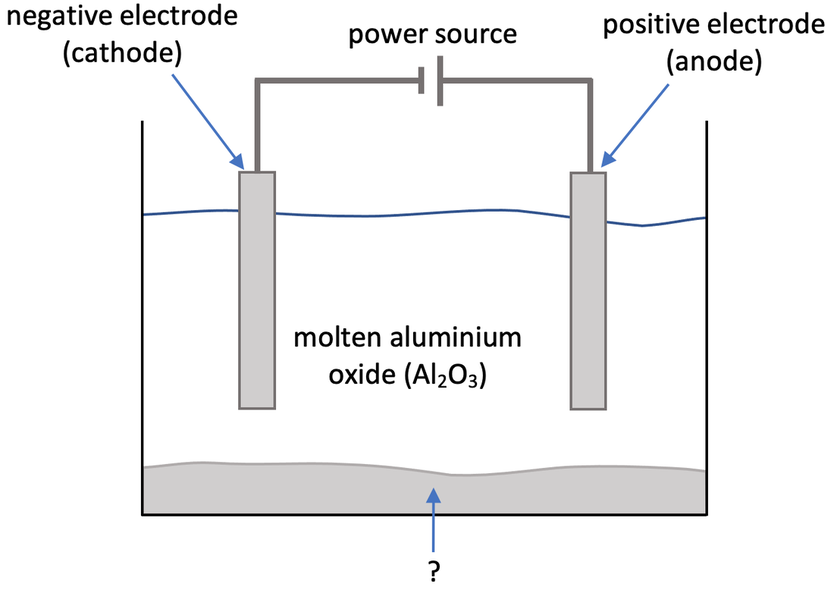
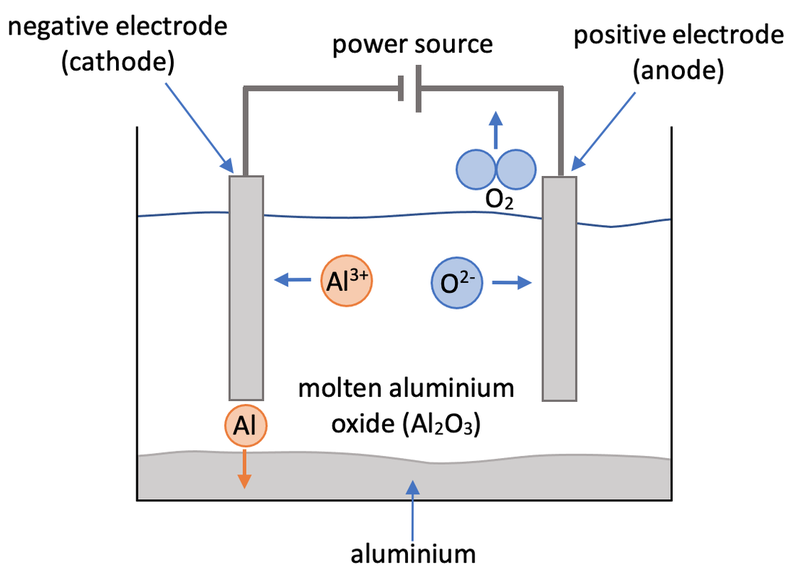
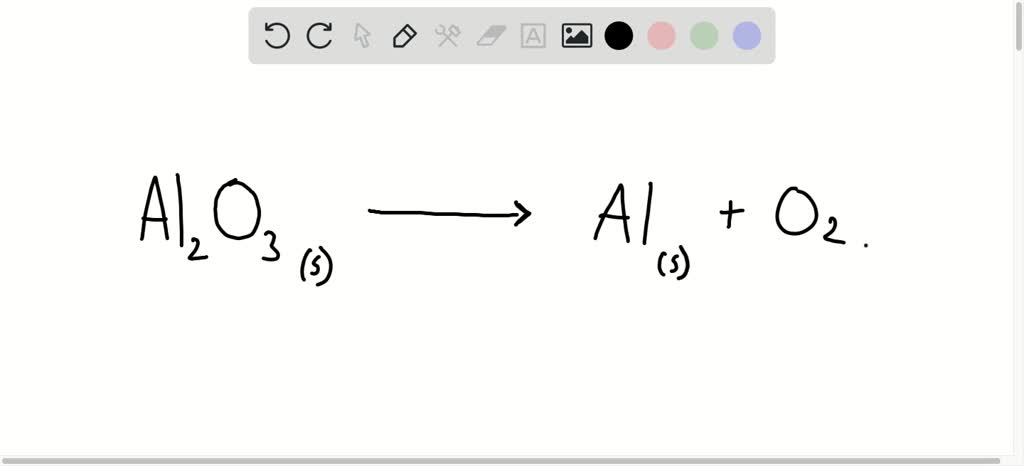
Construct and annotate a diagram of the electrolytic cell for the electrolysis of molten aluminium oxide

Principles of Water Electrolysis and Recent Progress in Cobalt‐, Nickel‐, and Iron‐Based Oxides for the Oxygen Evolution Reaction - Yu - 2022 - Angewandte Chemie International Edition - Wiley Online Library

Enhancing the Overall Electrocatalytic Water-Splitting Efficiency of Mo2C Nanoparticles by Forming Hybrids with UiO-66 MOF | ACS Omega

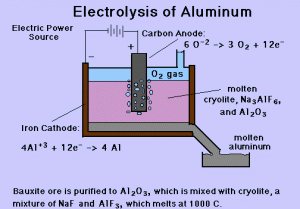
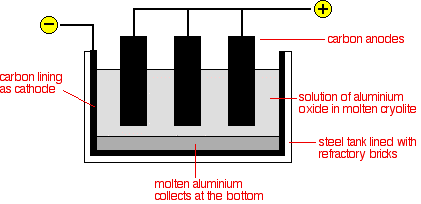
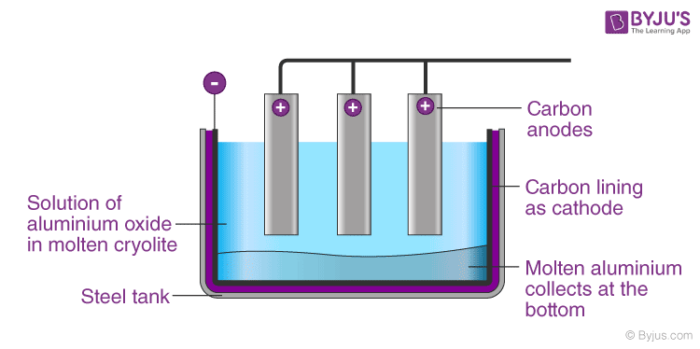
Extraction of aluminium aluminum recycling sodium by electrolysis raw materials bauxite rock salt electrode equations description of processs revision gcse igcse O Level KS4 science chemistry revision notes revising
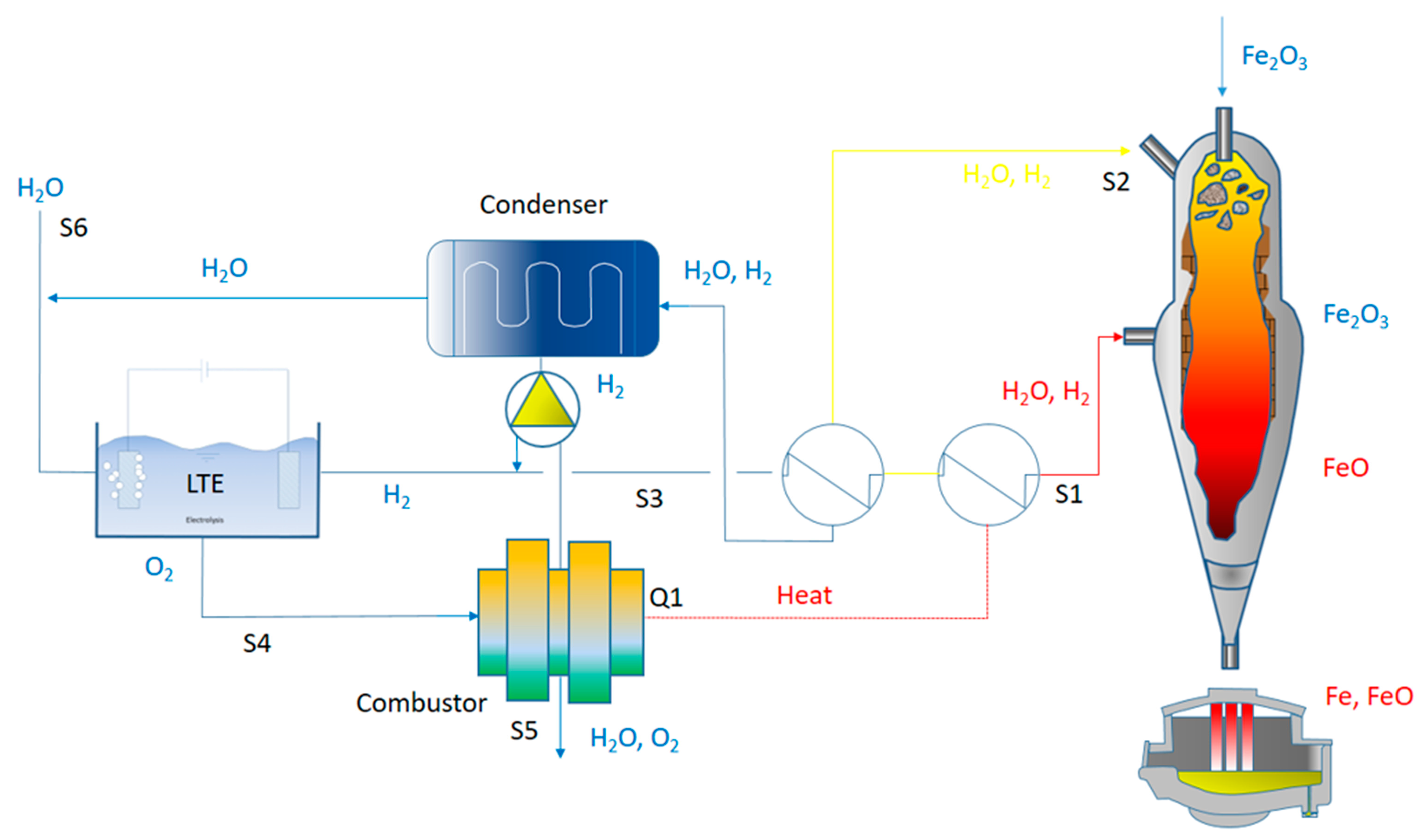
Metals | Free Full-Text | Water Electrolysis for the Production of Hydrogen to Be Employed in the Ironmaking and Steelmaking Industry
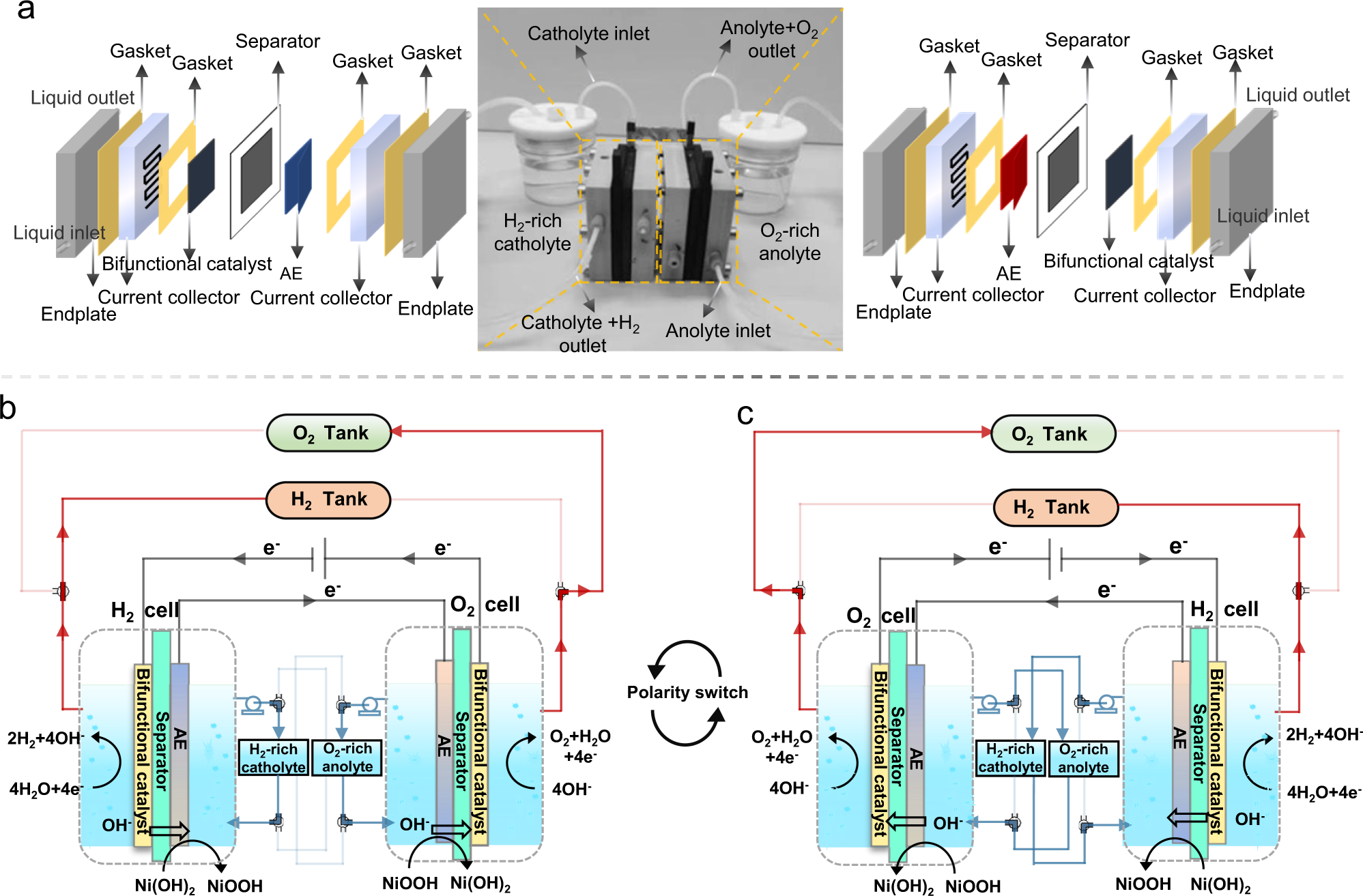
A membrane-free flow electrolyzer operating at high current density using earth-abundant catalysts for water splitting | Nature Communications

In the extraction of aluminium, the aluminium oxide for the electrolytic extraction of aluminium is obtained by heating aluminium hydroxide. Write the equation for this reaction.
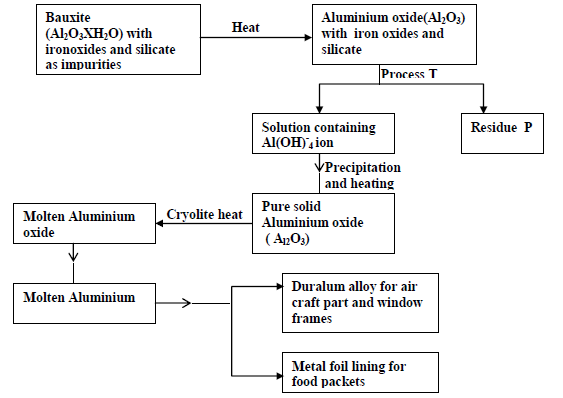
When 31.2g of hydrated. Aluminium oxide ( Al<sub>2O</sub>3</sub>XH<sub>2</sub>O) was heated to a constant mass of 20.6g of Aluminium oxide ( Al<sub>2</sub>O<sub>3</sub>) was obtained. Determine the value...