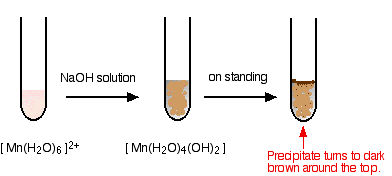
Chlorine is prepared in the laboratory by treating manganese dioxide(MnO2) with aqueous hydrochloric acid according to the reaction: 4HCl(aq) + MnO2(s)→ 2H2O(l) + MnCl2(aq) + Cl2(g) The amount of HCl that react
a) Two reagents that can be used to prepare chlorine gas are manganese (IV) oxide and concentrated - Tutorke
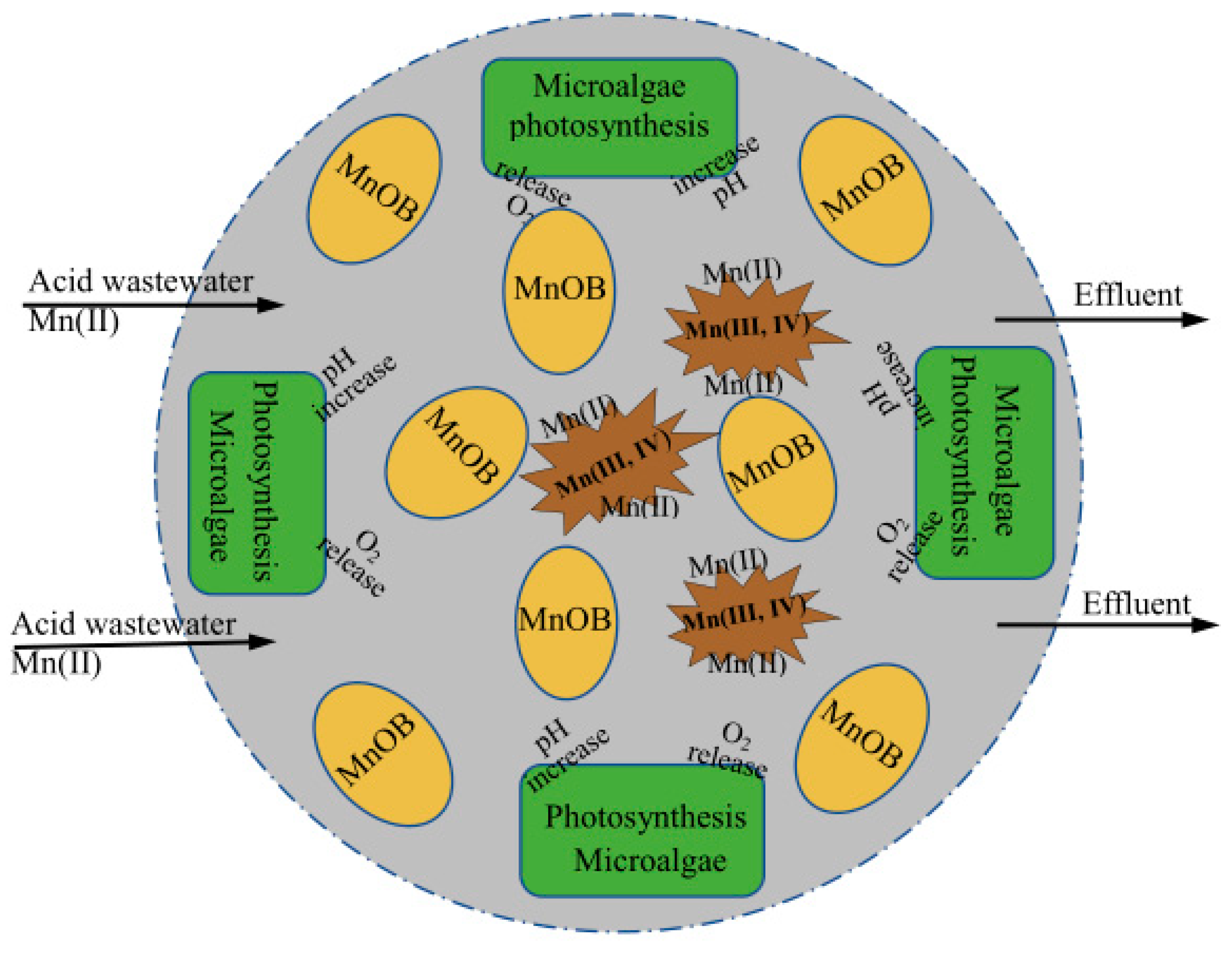
Water | Free Full-Text | Removal of Manganese(II) from Acid Mine Wastewater: A Review of the Challenges and Opportunities with Special Emphasis on Mn-Oxidizing Bacteria and Microalgae

Write the skeleton equations for the following chemical reactions and balance them by oxidation number method - CBSE Class 11 Chemistry - Learn CBSE Forum

Write the skeleton equations for the following chemical reactions and balance them by oxidation number method - CBSE Class 11 Chemistry - Learn CBSE Forum

Write the skeleton equation for the following chemical reaction and balance them by oxidation number method : (





![Solved Which of the following pairs is incorrect? [A] MnOz. | Chegg.com Solved Which of the following pairs is incorrect? [A] MnOz. | Chegg.com](https://media.cheggcdn.com/media%2F463%2F4635aaba-9ec2-4d99-895e-79512867e535%2Fimage.png)