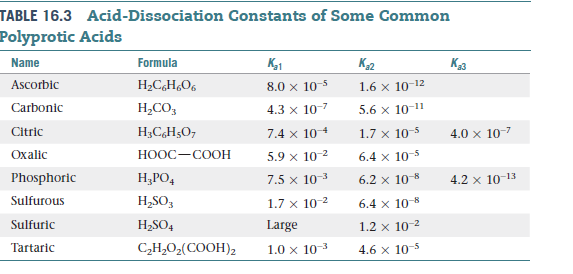
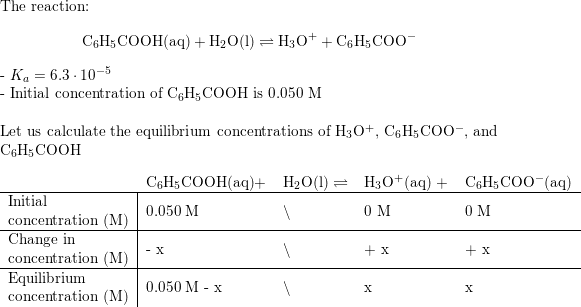Acid Dissociation Constant. Dissociation Constants For a generalized acid dissociation, the equilibrium expression would be This equilibrium constant. - ppt download

Dissociation Constants of Perchloric and Sulfuric Acids in Aqueous Solution | The Journal of Physical Chemistry B
![Weak Acids & Bases Chapter 16. Dissociation Constants Since weak acids do not dissociate completely, [H 3 O + ] ≠ [acid] For a generalized acid dissociation, - ppt download Weak Acids & Bases Chapter 16. Dissociation Constants Since weak acids do not dissociate completely, [H 3 O + ] ≠ [acid] For a generalized acid dissociation, - ppt download](https://images.slideplayer.com/33/9517443/slides/slide_2.jpg)
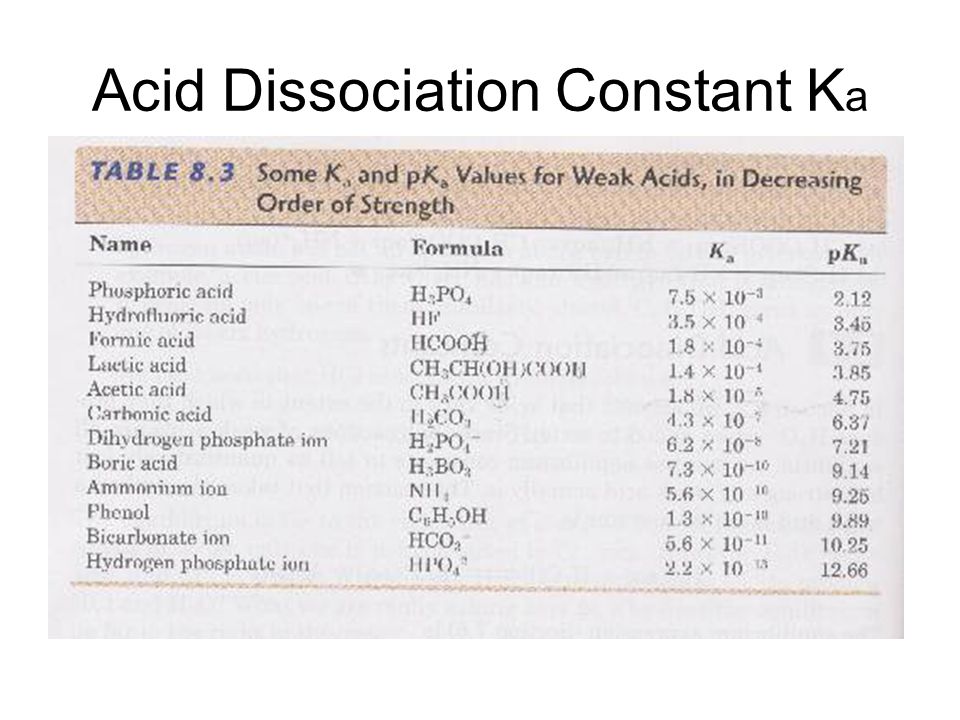
Weak Acids & Bases Chapter 16. Dissociation Constants Since weak acids do not dissociate completely, [H 3 O + ] ≠ [acid] For a generalized acid dissociation, - ppt download

Question Video: Calculating the Degree of Dissociation of a Solution of Phenol Given the Acid Dissociation Constant | Nagwa

equilibrium - How to calculate the dissociation constant of a weak acid from the titration with a strong base? - Chemistry Stack Exchange