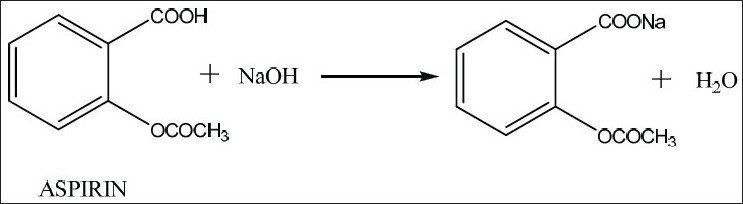
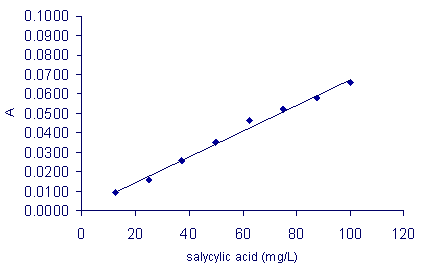
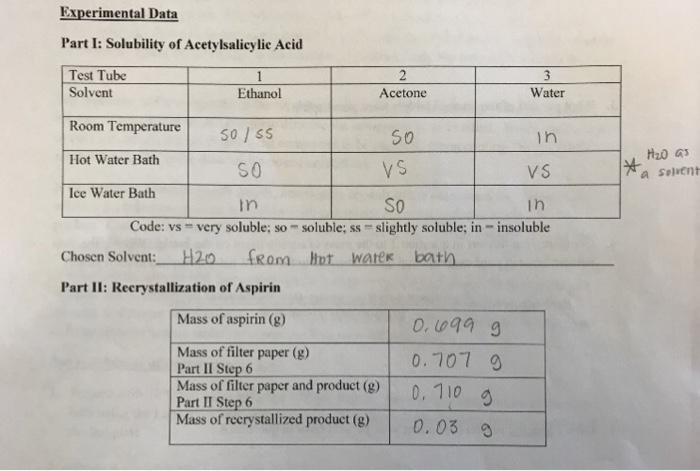
SOLVED: Find ' the solubility of aspirin in water and ethanol, Saponification is process where esters produce soap. Complete the react with strong following = base and is usually used to process;

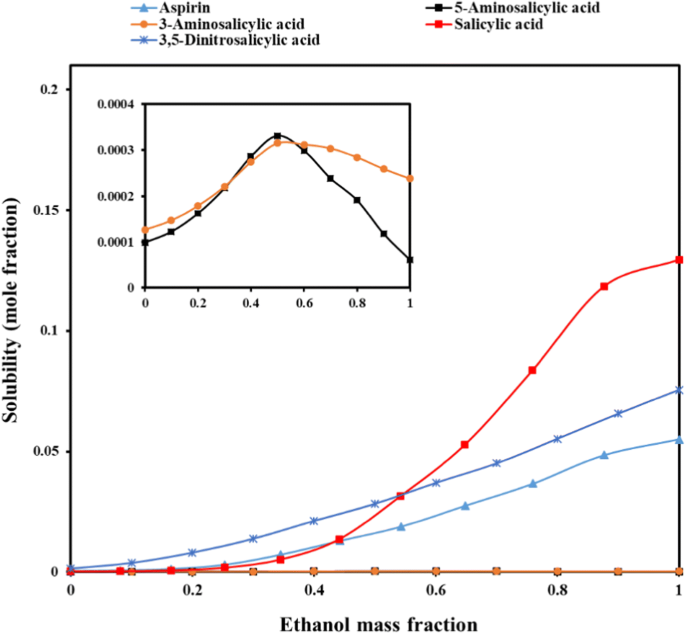
Solubility Study of Acetylsalicylic Acid in Ethanol + Water Mixtures: Measurement, Mathematical Modeling, and Stability Discussion | SpringerLink
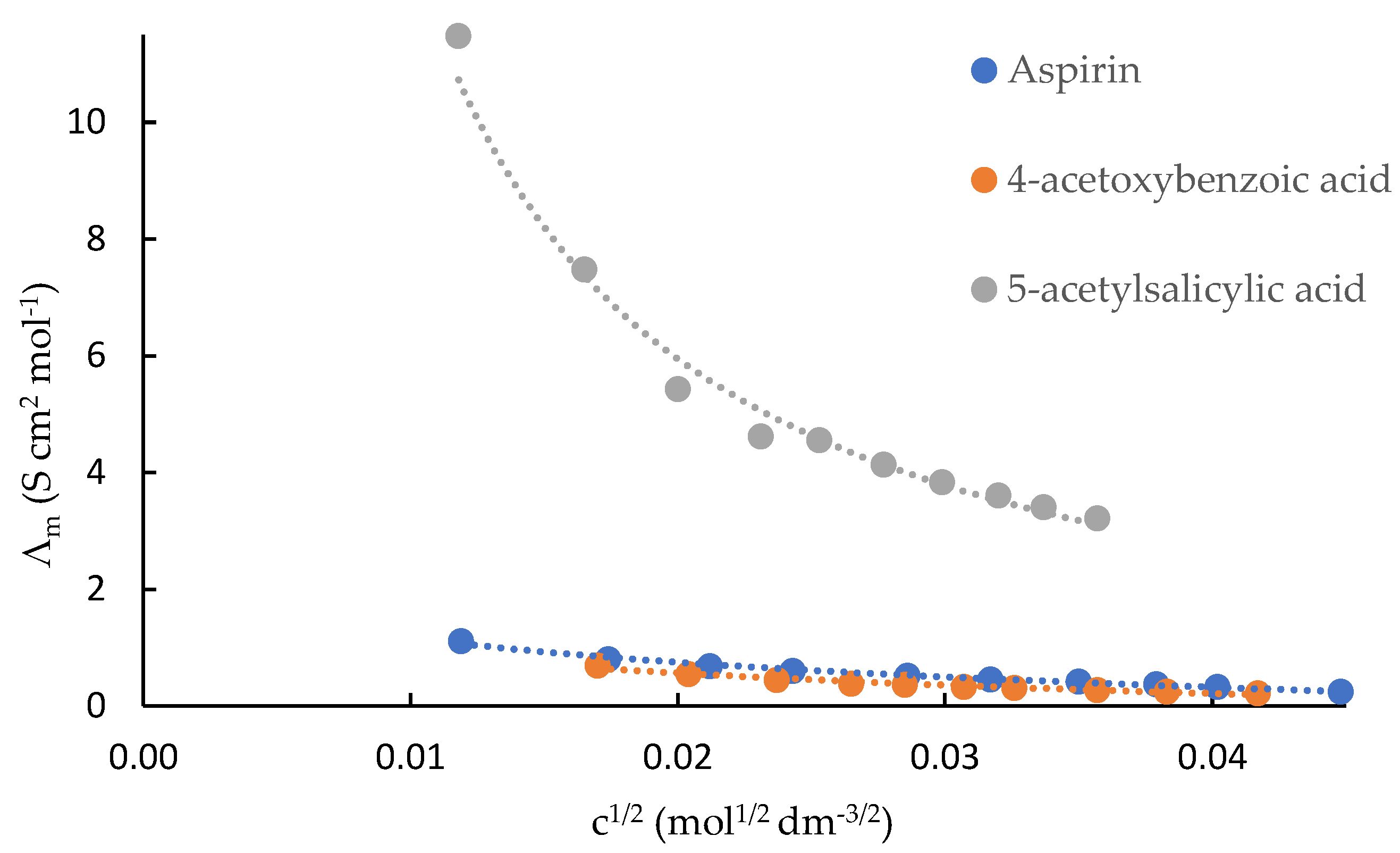
IJMS | Free Full-Text | From Solution Studies of Pharmaceuticals (Aspirin and Related Compounds) to the Thermodynamics of Aspirin-β-Cyclodextrin Interaction in water and N,N-Dimethylformamide
![PDF] Solubility Study of Acetylsalicylic Acid in Ethanol + Water Mixtures: Measurement, Mathematical Modeling, and Stability Discussion | Semantic Scholar PDF] Solubility Study of Acetylsalicylic Acid in Ethanol + Water Mixtures: Measurement, Mathematical Modeling, and Stability Discussion | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4365afd879faf3d46cb23349ba7518f59178f8cb/7-Figure4-1.png)
PDF] Solubility Study of Acetylsalicylic Acid in Ethanol + Water Mixtures: Measurement, Mathematical Modeling, and Stability Discussion | Semantic Scholar
Solubility Study of Acetylsalicylic Acid in Ethanol + Water Mixtures: Measurement, Mathematical Modeling, and Stability Discussion | SpringerLink

Influence of polymeric excipients on the solubility of aspirin: Experimental measurement and model prediction - ScienceDirect
Between acetylsalicylic acid and benzoic acid, which is most polar if you look only at the structure (not at solubility in water g/l), and why? Why is that structure most polar? -

Solubility of salicylic acid in ethanol, propylene glycol, and N-methyl-2-pyrrolidone at various temperatures and their binary mixtures at 298.2 K - ScienceDirect

Solubility of Acetylsalicylic Acid in Ethanol, Acetone, Propylene Glycol, and 2-Propanol | Journal of Chemical & Engineering Data

Solubility of salicylic acid in ethanol, propylene glycol, and N-methyl-2-pyrrolidone at various temperatures and their binary mixtures at 298.2 K | Semantic Scholar

Solubility study of acetylsalicylic acid in ethanol + water mixtures: measurement, mathematical modeling, and stability discussion