![The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ] The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]](https://i.ytimg.com/vi/AufT6_CoFWY/maxresdefault.jpg)
The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]
The pH of an acetic acid solution is 3.26. What is the concentration of acetic acid and what is the percent of acid that's ionized? - Quora
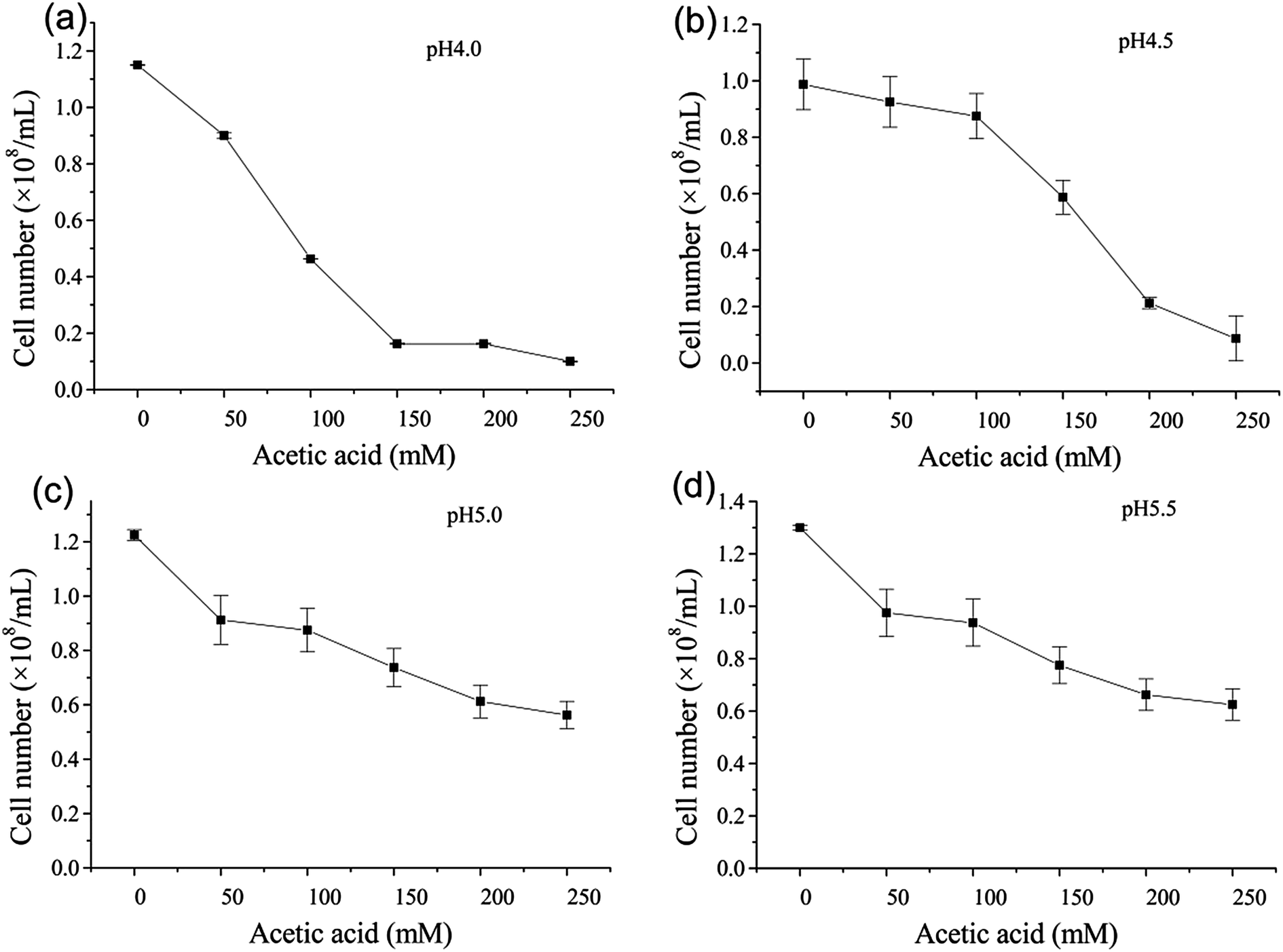
Control of pH by acetic acid and its effect on ethanol fermentation in an integrated ethanol–methane fermentation process - RSC Advances (RSC Publishing) DOI:10.1039/C6RA04129A
![PDF] Standards for pH Measurements of Bis-((2-Hydroxyethyl)amino)acetic Acid (BICINE) from (278.15 to 328.15) K | Semantic Scholar PDF] Standards for pH Measurements of Bis-((2-Hydroxyethyl)amino)acetic Acid (BICINE) from (278.15 to 328.15) K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0485e11983e6b106e34893763c3fb7dcf6fe728b/2-Table1-1.png)
PDF] Standards for pH Measurements of Bis-((2-Hydroxyethyl)amino)acetic Acid (BICINE) from (278.15 to 328.15) K | Semantic Scholar

The Ka value for acetic acid, CH3COOH(aq), is 1.8x10^-5. Calculate the ph of a 2.80 M acetic acid solution - Home Work Help - Learn CBSE Forum

pH calculations and more in fundamentals of pharmaceutics. : Calculate pH of 100 ml buffer solution containing 0.1 g acetic acid and 0.2 g sodium actetate.
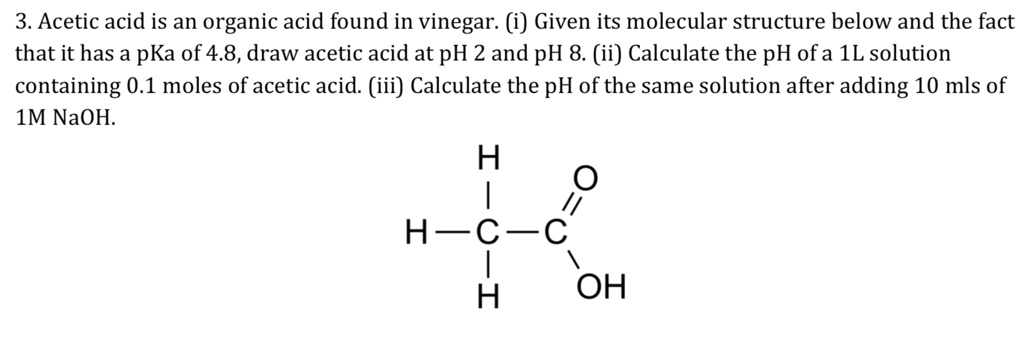
SOLVED: 3. Acetic acid is an organic acid found in vinegar: (i) Given its molecular structure below and the fact that it has a pKa of 4.8,draw acetic acid at pH 2
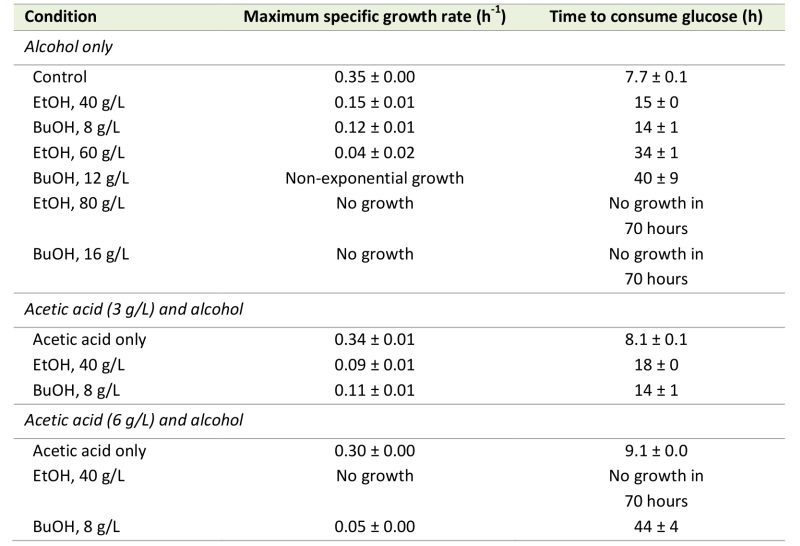
Alcohols enhance the rate of acetic acid diffusion in S. cerevisiae: biophysical mechanisms and implications for acetic acid tolerance

![Sample Paper] Anita added a drop each of diluted acetic acid and Sample Paper] Anita added a drop each of diluted acetic acid and](https://d1avenlh0i1xmr.cloudfront.net/aec8a3c5-7906-481e-894e-29e9a7a4be11/ph-scale-techoo.jpg)