EP0124911B1 - 4-oxo-thiazolidin-2-yliden-acetamide derivatives, process for their preparation, and medicaments for the treatment of central nervous system illnesses containing them - Google Patents

Figure 1 | Determination of Reactive Carbonyl Species, Which Mediate Reactive Oxygen Species Signals in Plant Cells | SpringerLink

A New Method for the One-Step Synthesis of α,β-Unsaturated Carbonyl Systems from Saturated Alcohols and Carbonyl Compounds | Journal of the American Chemical Society

A New Method for the One-Step Synthesis of α,β-Unsaturated Carbonyl Systems from Saturated Alcohols and Carbonyl Compounds | Journal of the American Chemical Society
![An unsaturated hydrocarbon X absorbs two hydrogen molecules on catalytic hydrogenation, and also gives following reaction : X [ Zn/H2O ]O3 A [ ] [ Ag (NH3)2 ]^+ B (3 - oxo - hexanedicarboxylic acid) X will be : An unsaturated hydrocarbon X absorbs two hydrogen molecules on catalytic hydrogenation, and also gives following reaction : X [ Zn/H2O ]O3 A [ ] [ Ag (NH3)2 ]^+ B (3 - oxo - hexanedicarboxylic acid) X will be :](https://haygot.s3.amazonaws.com/questions/1476524_1697399_ans_5f7adeae8b3c4be6afe34dceb75f4554.png)
An unsaturated hydrocarbon X absorbs two hydrogen molecules on catalytic hydrogenation, and also gives following reaction : X [ Zn/H2O ]O3 A [ ] [ Ag (NH3)2 ]^+ B (3 - oxo - hexanedicarboxylic acid) X will be :

The mechanism of the reaction of hydrazines with α,β-unsaturated carbonyl compounds to afford hydrazones and 2-pyrazolines (4,5-dihydro-1H-pyrazoles): Experimental and theoretical results - ScienceDirect

Examples of α,β-unsaturated aldehydes, ketones and nitro compounds that... | Download Scientific Diagram
Analytics for US Patent No. 6969770, 2-oxo-1-pyrrolidine derivatives, process for preparing them and their uses
![IJMS | Free Full-Text | Synthesis and Antimicrobial Evaluation of Some Novel Bis-α,β-Unsaturated Ketones, Nicotinonitrile, 1,2-Dihydropyridine-3-carbonitrile, Fused Thieno[2,3-b]pyridine and Pyrazolo[3,4-b]pyridine Derivatives IJMS | Free Full-Text | Synthesis and Antimicrobial Evaluation of Some Novel Bis-α,β-Unsaturated Ketones, Nicotinonitrile, 1,2-Dihydropyridine-3-carbonitrile, Fused Thieno[2,3-b]pyridine and Pyrazolo[3,4-b]pyridine Derivatives](https://www.mdpi.com/ijms/ijms-14-02967/article_deploy/html/images/ijms-14-02967f2.png)
IJMS | Free Full-Text | Synthesis and Antimicrobial Evaluation of Some Novel Bis-α,β-Unsaturated Ketones, Nicotinonitrile, 1,2-Dihydropyridine-3-carbonitrile, Fused Thieno[2,3-b]pyridine and Pyrazolo[3,4-b]pyridine Derivatives
Molecules | Free Full-Text | The Heck Reaction Applied to 1,3- and 1,2- Unsaturated Derivatives, a Way towards Molecular Complexity
![PDF) Highly regio- and diastereoselective synthesis of oxo-1,2 ,3,4-tetrahydropyrazino[1,2-a]indoles, based on a post-Ugi condensation joint experimental and computational study | Morteza Shiri - Academia.edu PDF) Highly regio- and diastereoselective synthesis of oxo-1,2 ,3,4-tetrahydropyrazino[1,2-a]indoles, based on a post-Ugi condensation joint experimental and computational study | Morteza Shiri - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/65531555/mini_magick20210127-22542-tr1dz8.png?1611759983)
PDF) Highly regio- and diastereoselective synthesis of oxo-1,2 ,3,4-tetrahydropyrazino[1,2-a]indoles, based on a post-Ugi condensation joint experimental and computational study | Morteza Shiri - Academia.edu

Figure 1 from Atmospheric Chemistry of Unsaturated Carbonyls: Butenedial, 4- Oxo-2-pentenal, 3-Hexene-2,5-dione, Maleic Anhydride, 3H-Furan-2-one, and 5-Methyl-3H-furan-2-one. | Semantic Scholar
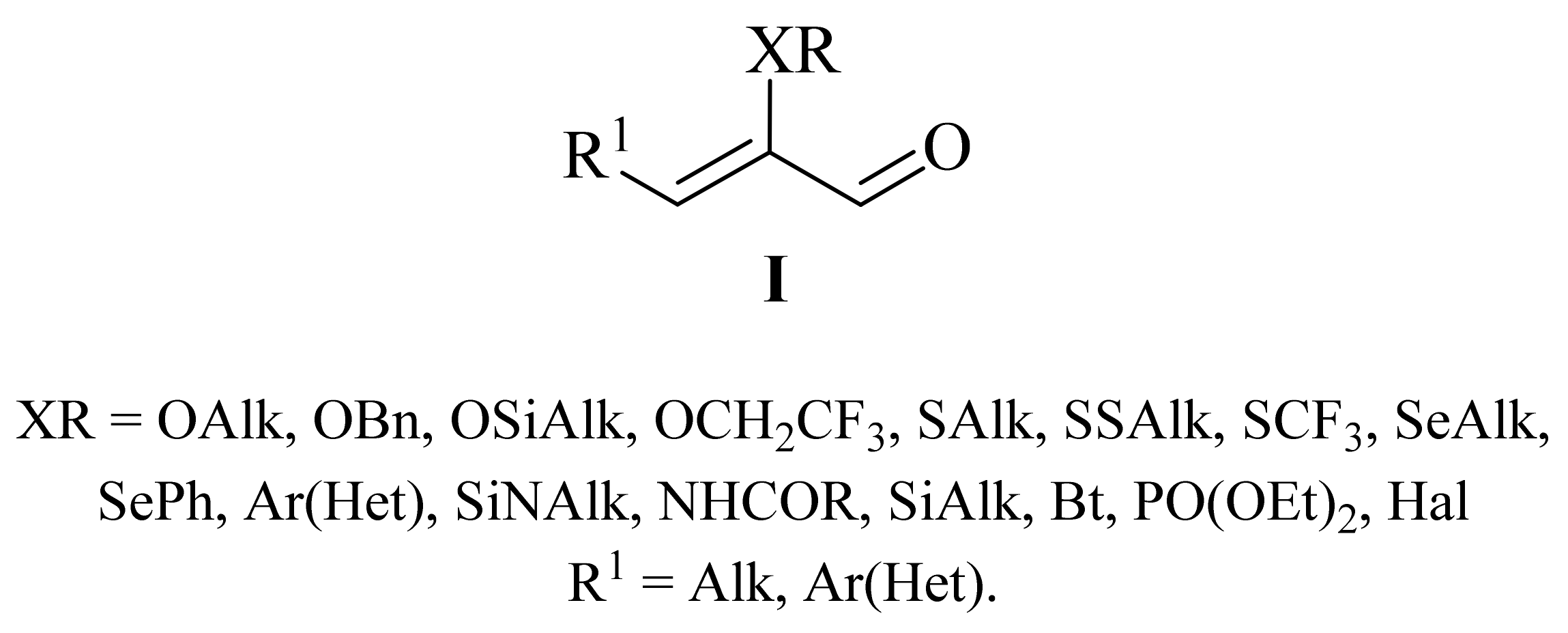
Molecules | Free Full-Text | α-Functionally Substituted α,β-Unsaturated Aldehydes as Fine Chemicals Reagents: Synthesis and Application
![Brønsted Base-Catalyzed Domino Annulation of α-oxo-β, γ-Unsaturated Ketones and Malononitrile: Facile Access to Poly-substituted Tetrahydrocyclopenta[b]furanols | Organic Chemistry | ChemRxiv | Cambridge Open Engage Brønsted Base-Catalyzed Domino Annulation of α-oxo-β, γ-Unsaturated Ketones and Malononitrile: Facile Access to Poly-substituted Tetrahydrocyclopenta[b]furanols | Organic Chemistry | ChemRxiv | Cambridge Open Engage](https://chemrxiv.org/engage/api-gateway/chemrxiv/assets/orp/resource/item/636c69f7c4c8790ef724762b/largeThumb/construction-of-tetrahydrocyclopenta-b-furanols-via-br-nsted-base-catalyzed-annulation-reactions.jpg)
Brønsted Base-Catalyzed Domino Annulation of α-oxo-β, γ-Unsaturated Ketones and Malononitrile: Facile Access to Poly-substituted Tetrahydrocyclopenta[b]furanols | Organic Chemistry | ChemRxiv | Cambridge Open Engage
![A new entrant in ketene family: Generation of 2‐(3‐Oxo‐allyl)‐isoindole‐1,3‐dione and their [4+2] Cycloadditions with 1,3‐Diazabuta‐1,3‐dienes | GDCh.app A new entrant in ketene family: Generation of 2‐(3‐Oxo‐allyl)‐isoindole‐1,3‐dione and their [4+2] Cycloadditions with 1,3‐Diazabuta‐1,3‐dienes | GDCh.app](https://onlinelibrary.wiley.com/cms/asset/9978a6d3-cd22-40cf-bbae-9e75af1894df/ajoc202200636-toc-0001-m.png)